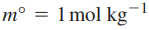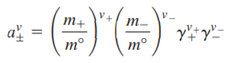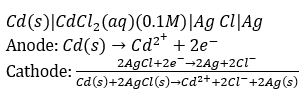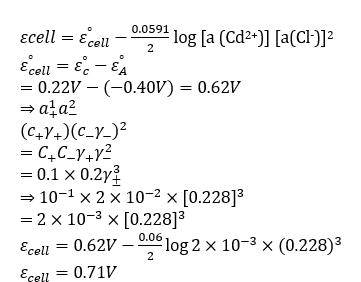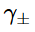Correct option is A
Nernst equation
In electrochemistry, the Nernst equation is a chemical thermodynamical relationship that permits the calculation of the reduction potential of a reaction (half-cell or full cell reaction) from the standard electrode potential, absolute temperature, the number of electrons involved in the redox reaction, and activities (often approximated by concentrations) of the chemical species undergoing reduction and oxidation respectively.

Activities and Activity Coefficients for Electrolyte Solutions
Consider the Gibbs energy of the solution, which can be written as
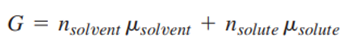
For the general electrolyte
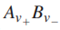

that dissociates completely, one can also write an equivalent expression for G:
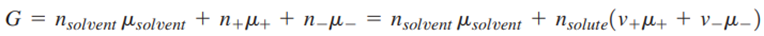
where
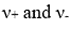
are the stoichiometric coefficients of the cations and anions, respectively, produced upon dissociation of the electrolyte. In shorthand notation, an electrolyte is called a 1-1 electrolyte if
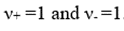
Therefore,
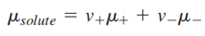
Although this equation is formally correct for a strong electrolyte, one can never make a solution of either cations or anions alone, because any solution is electrically neutral. Therefore, it is useful to define a mean ionic chemical potential
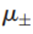
for the solute
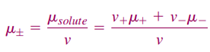
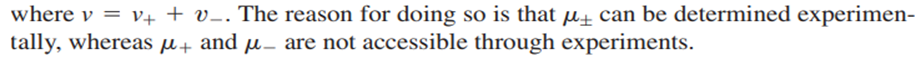
The next task is to relate the chemical potentials of the solute and its individual ions to the activities of these species. For the individual ions,
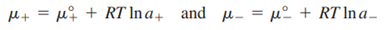
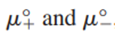
are the standard chemical potentials of the ions. An equation for the mean ionic chemical potential is obtained as
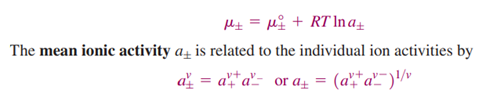
If the ionic activities are referenced to the concentration units of molality, then
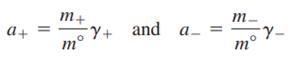
Where
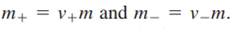
Because the activity is unitless, the molality must be referenced to a standard state concentration chosen to be