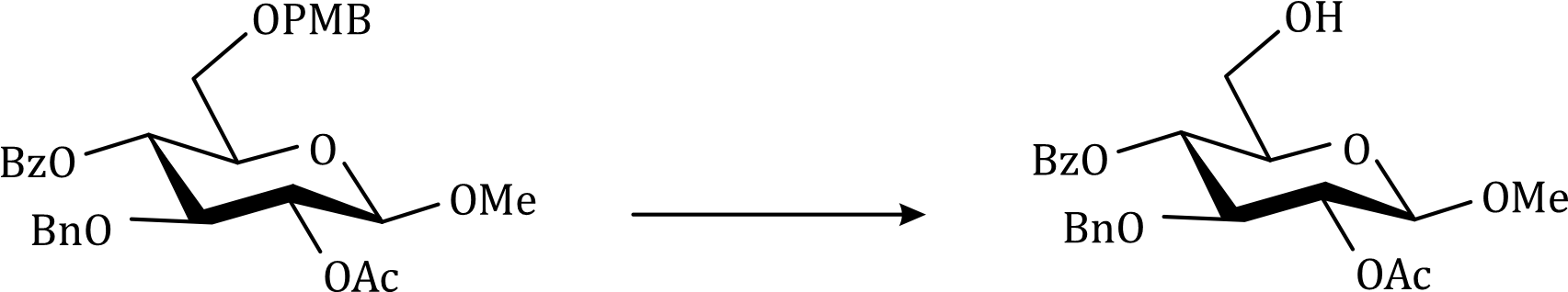
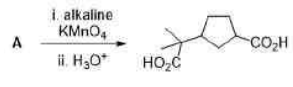
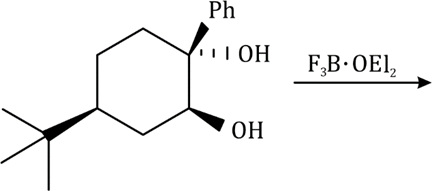
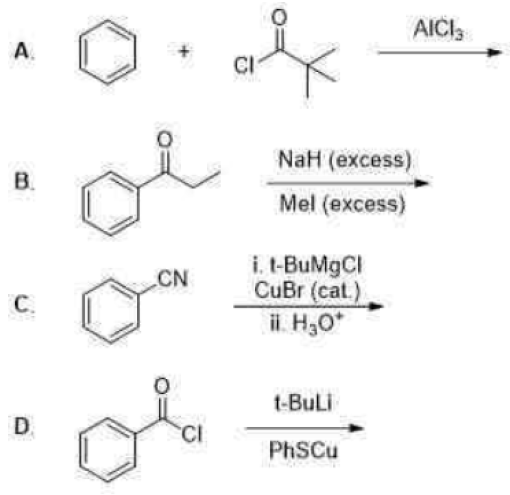
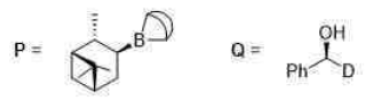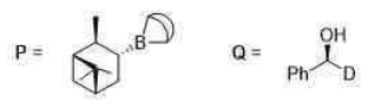Correct option is B
The hydration of alkenes via hydroboration-oxidation provides a valuable tool for the synthesis of a wide variety of alcohols of predictable regio- and stereochemistry.
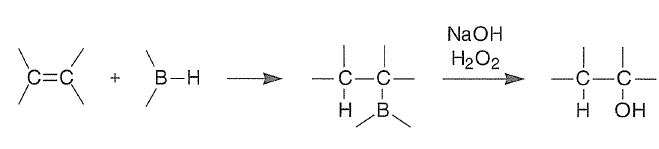
Borane-tetrahydrofuran (BH3.THF) and borane dimethyl sulfide (BH3.SMe2) are the reagents of choice for hydroboration of alkenes.
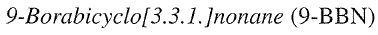
is available by hydroboration of 1,5-cyclooctadiene with one equivalent of BH3, or commercially either as a crystalline dimer or as a THF solution. It is frequently used as an anchor group in organoborane reactions, allowing an efficient utilization of valuable alkenes.
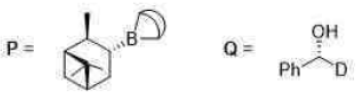
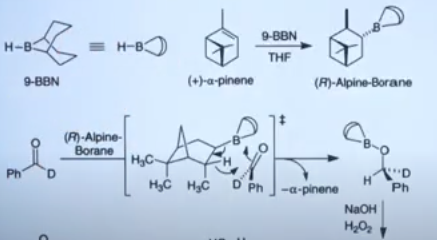
Alpine-Borane, prepared by hydroboration of α-pinene with
reduces aldehyde, α-keto esters and acetylenic ketones with excellent enantioselectivity. The reduction proceeds via a cyclic process.
Oxidation of Organoboranes. The facile reactions of olefins and dienes with various hydroborating agents makes a variety of organoboranes readily available. Organoboranes tolerate many functional groups and are formed in a stereospecific manner. The boron atom in these organoboranes can be readily substituted with a variety of functional groups. For example, hydroboration followed by in situ oxidation by alkaline hydrogen peroxide provides for the anti-Markovnikov hydration of double bonds.
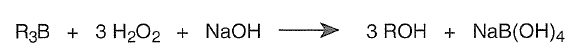
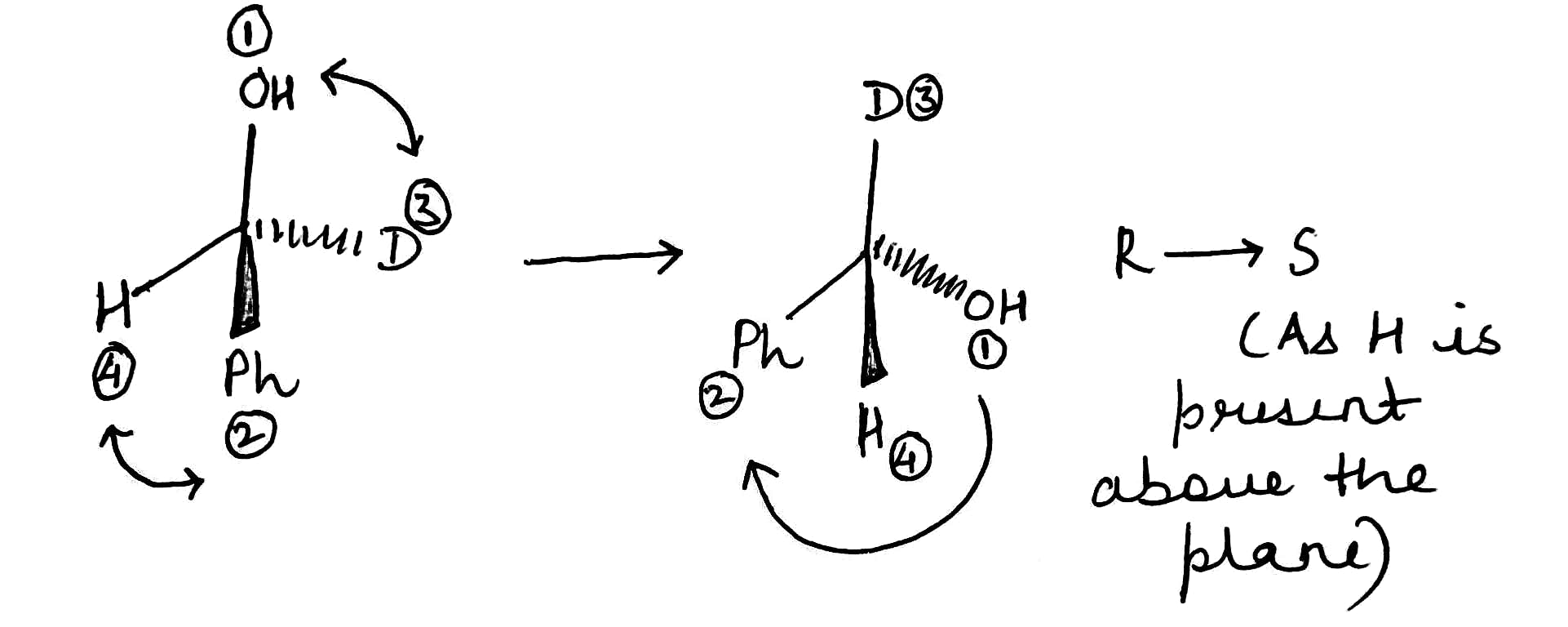
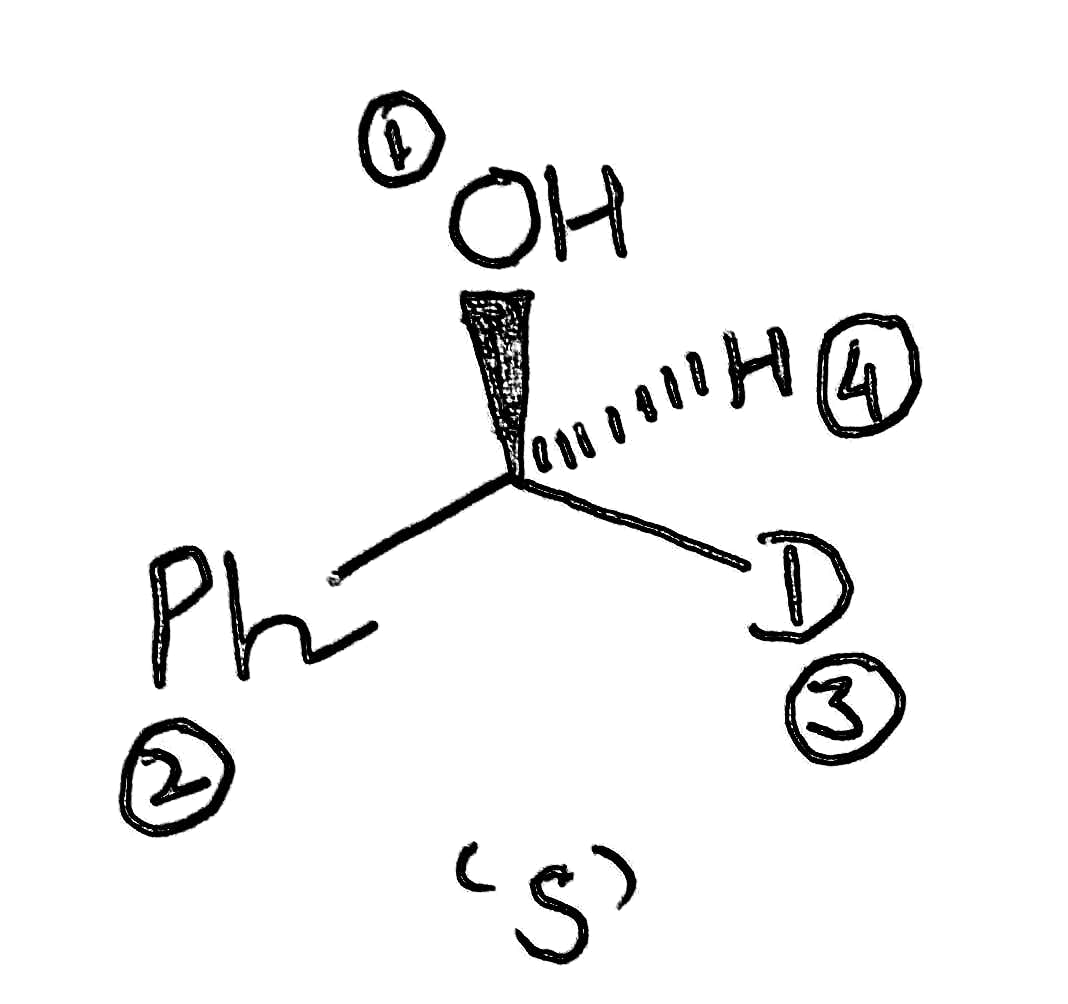
The product has S configuration.
The image shown below (option B) has S configuration.
Information Booster
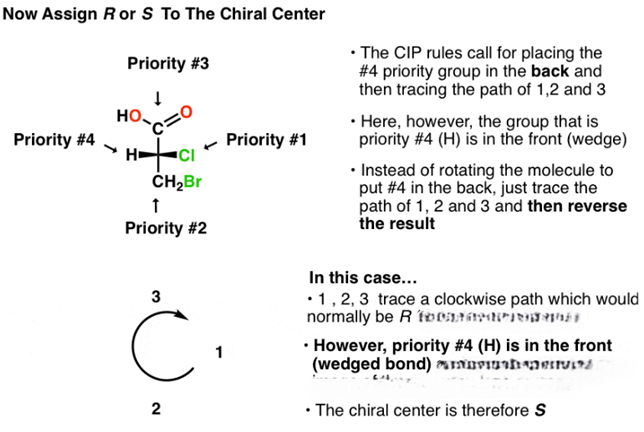
The R/S system is an important nomenclature system for denoting enantiomers. This approach labels each chiral center R or S according to a system by which its substituents are each assigned a priority, according to the Cahn–Ingold–Prelog priority rules (CIP), based on atomic number. When the center is oriented so that the lowest-priority substituent of the four is pointed away from the viewer, the viewer will then see two possibilities: if the priority of the remaining three substituents decreases in clockwise direction, it is labeled R (for Latin: rectus – right); if it decreases in counterclockwise direction, it is S (for Latin: sinister – left).
The Cahn–Ingold–Prelog (CIP) sequence rules are a standard process to completely and unequivocally name a stereoisomer of a molecule. The purpose of the CIP system is to assign an R or S descriptor to each stereocenter.
1. Compare the atomic number (Z) of the atoms directly attached to the stereocenter; the group having the atom of higher atomic number Z receives higher priority (i.e. number 1).
2. If there is a tie, the atoms at distance 2 from the stereocenter have to be considered: a list is made for each group of further atoms bonded to the one directly attached to the stereocenter. Each list is arranged in order of decreasing atomic number Z. Then the lists are compared atom by atom; at the earliest difference, the group containing the atom of higher atomic number Z receives higher priority.