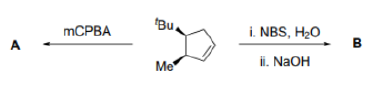
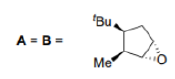
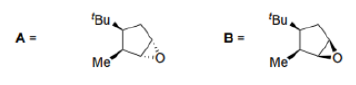
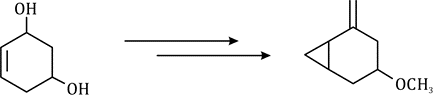
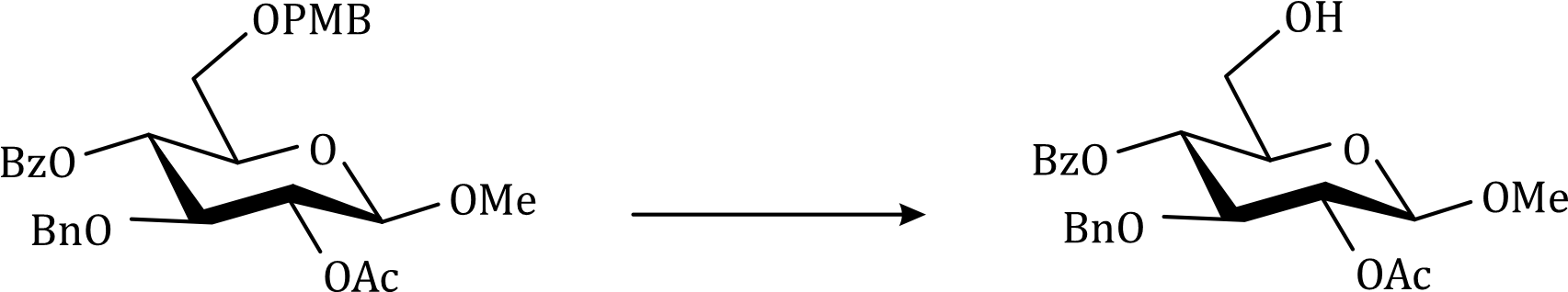
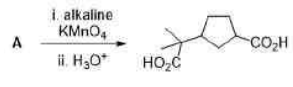
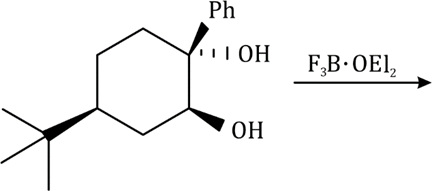
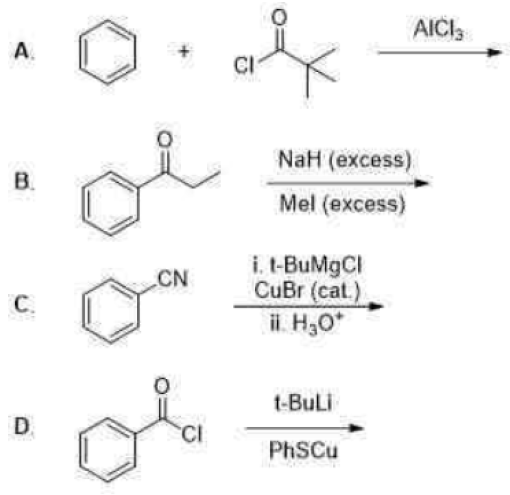
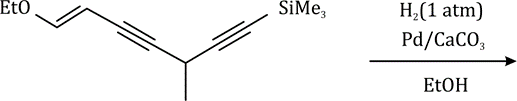Correct option is B
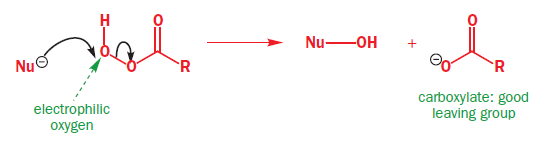
The most commonly used epoxidizing agents are peroxy-carboxylic acids. Peroxy-acids (or peracids) have an extra oxygen atom between the carbonyl group and their acidic hydrogen—they are half-esters of hydrogen peroxide (H2O2).They are rather less acidic than carboxylic acids because their conjugate base is no longer stabilized by delocalization into the carbonyl group reagent. But they are electrophilic at oxygen, because attack there by a nucleophile displaces carboxylate, a good leaving group. The LUMO of a peroxy-carboxylic acid is the
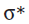
orbital of the weak O–O bond.
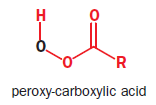
The most commonly used peroxy-acid is known as m-CPBA, or meta-ChloroPeroxyBenzoic Acid.
The alkene attacks the peroxy-acid from the centre of the HOMO, its π orbital.
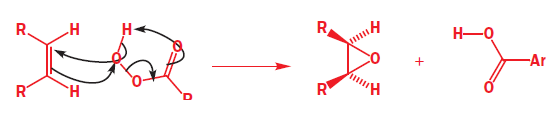
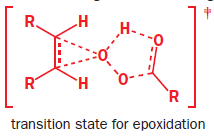
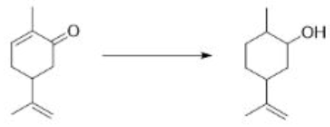
Start with the nucleophilic π bond: send the electrons on to oxygen, breaking O–O and forming a new carbonyl bond. Use those electrons to pick up the proton, and use the old O–H bond’s electrons to make the second new C–O bond. Because both new C–O bonds are formed on the same face of the alkene’s π bond, the geometry of the alkene is reflected in the stereochemistry of the epoxide. The reaction is therefore stereospecific.
In the product A, the epoxide will be formed from the less hindered side (below the plane).
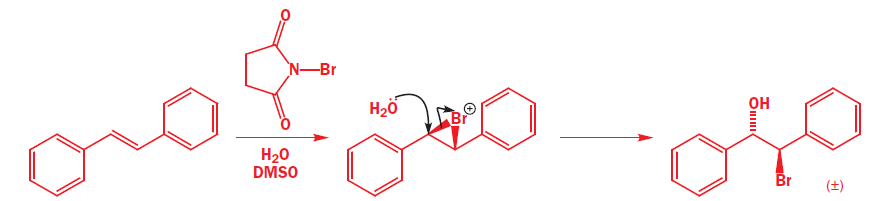
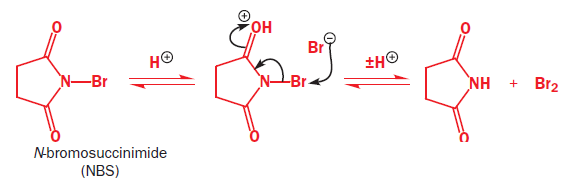
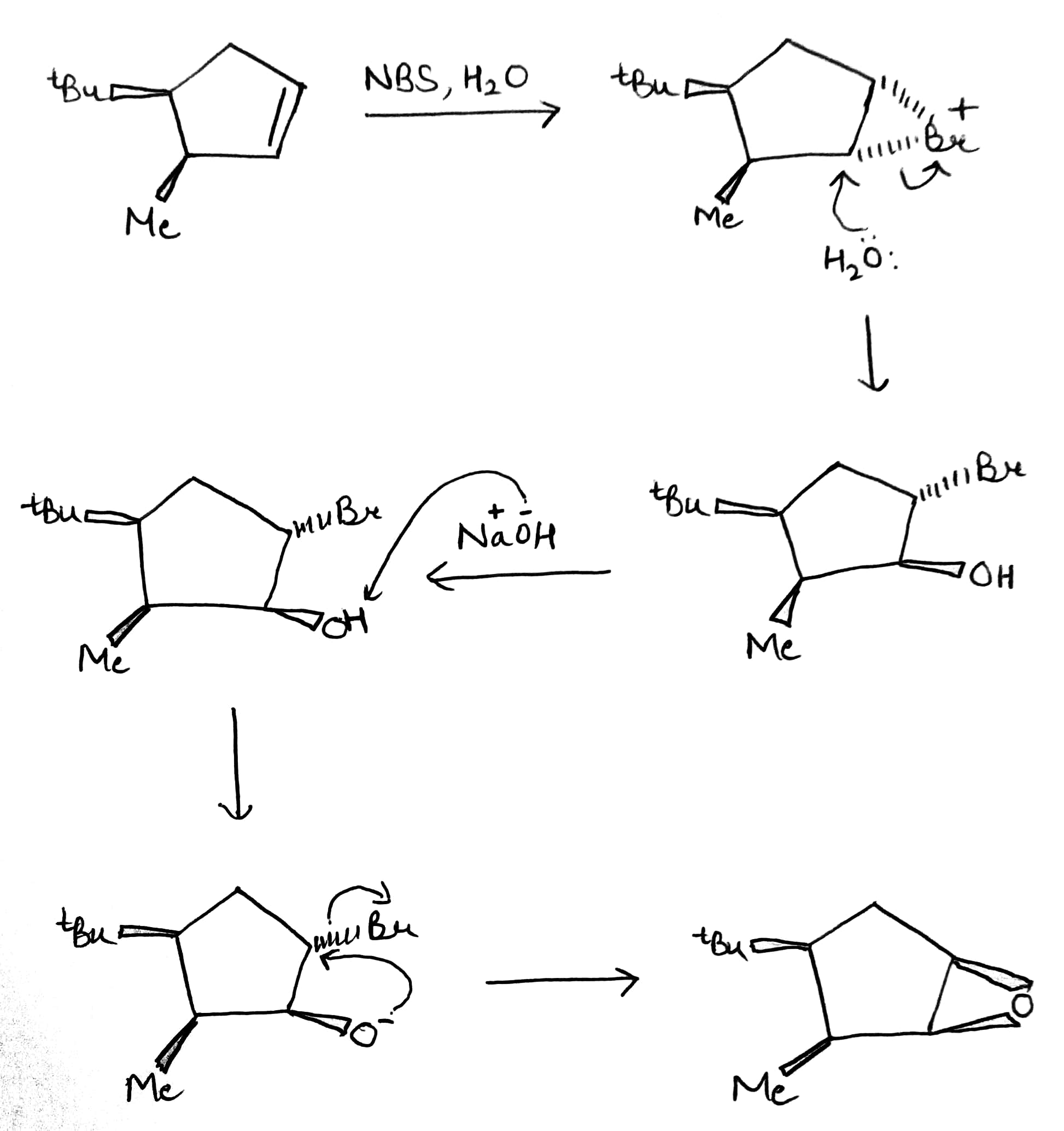
The reagent used to form the bromonium ion is called N-bromosuccinimide, or NBS for short. Unlike the noxious brown liquid bromine, NBS is an easily handled crystalline solid, and is perfect for electrophilic addition of bromine to alkenes when the bromonium ion is not intended to be opened by Br-.It works by providing a very small concentration of Br2 in solution: a small amount of HBr is enough to get the reaction going, and thereafter every addition reaction produces another molecules of HBr which liberates more Br2 from NBS. In a sense, NBS is a source of ‘Br+’.