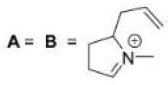
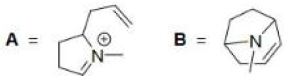
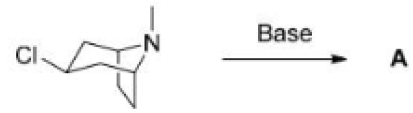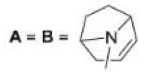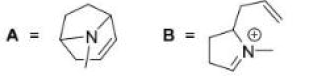Correct option is D
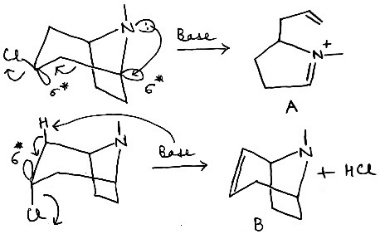
For the formation of the product B, the base abstracts a proton adjacent to the carbon bearing the chlorine, resulting in the elimination of HCl and the formation of a double bond within the bicyclic system.
For the formation of the product A, the lone pair of the nitrogen attacks the σ* of the C-C bond. The electrons in the C-C bond in turn enter the σ* of the C-Cl bond.
Intramolecular describes a process or characteristic limited within the structure of a single molecule, a property or phenomenon limited to the extent of a single molecule. In intramolecular organic reactions, two reaction sites are contained within a single molecule. This configuration elevates the effective concentration of the reacting partners resulting in high reaction rates.