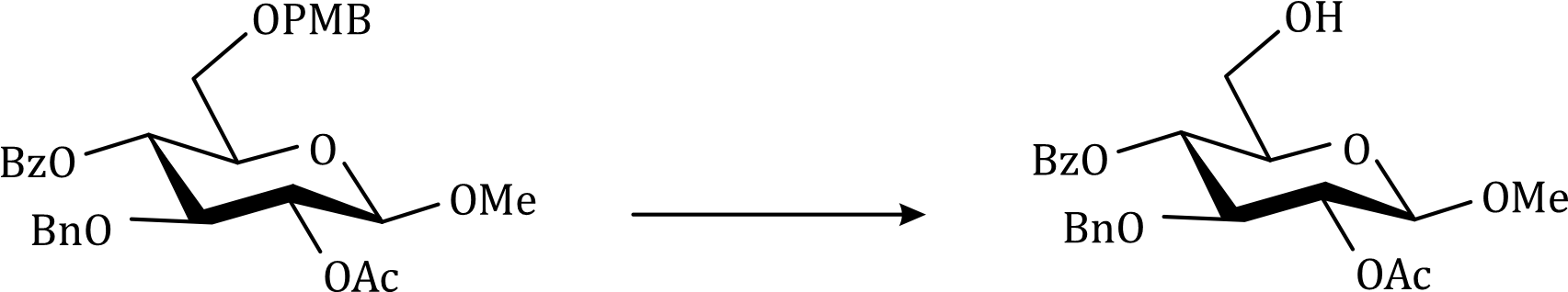
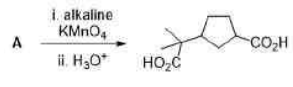
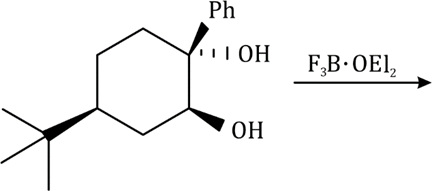
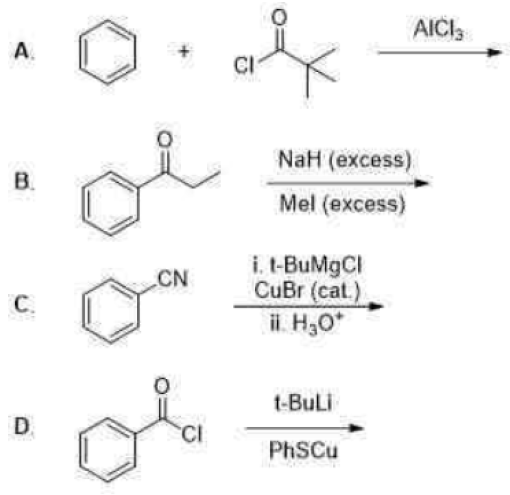
Correct option is C
Grignard reagents add directly to the carbonyl group of α,β-unsaturated aldehydes and ketones to give allylic alcohols. The hard Grignard reagent prefers to attack the harder C=O rather than the softer C=C electrophilic centre.
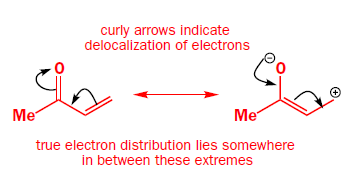
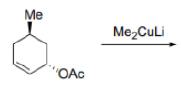
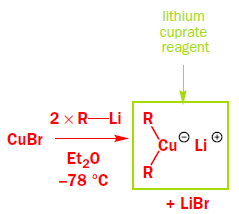
The copper works by transmetallating the Grignard reagent to give an organocopper reagent. Organocoppers are softer than Grignard reagents, and add in a conjugate fashion to the softer C=C double bond. Once the organocopper has added, the copper salt is available to transmetallate some more Grignard, and only a catalytic amount is required.
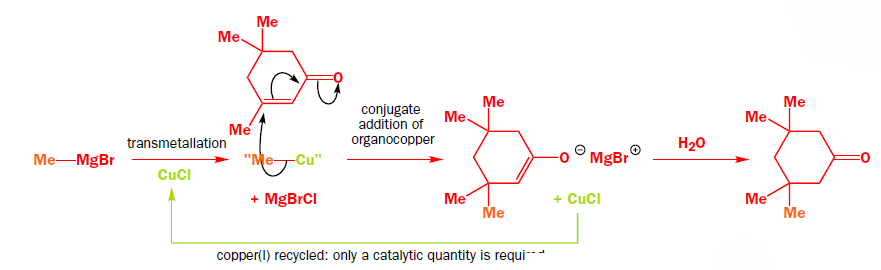
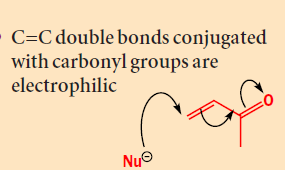
Addition of organocopper reagents to α,β-unsaturated carbonyl compounds (enones and conjugated esters) generates enolates with concomitant introduction of an organic group at the β-position.
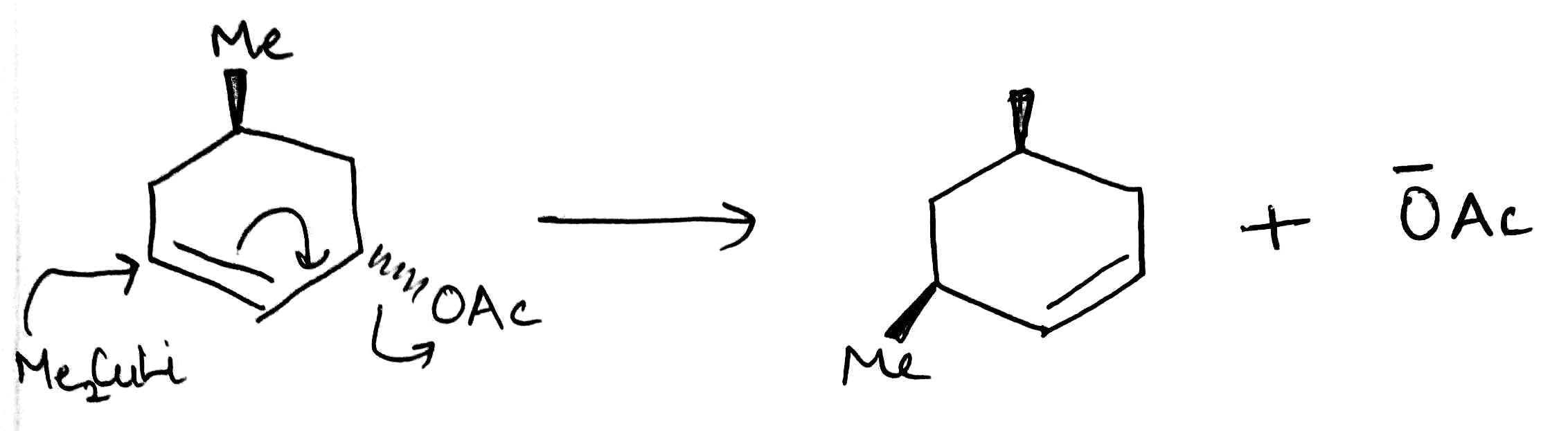
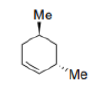
The reaction is also stereoselective in that introduction of the "Me" group occurs preferentially from the less hindered side of the molecule.