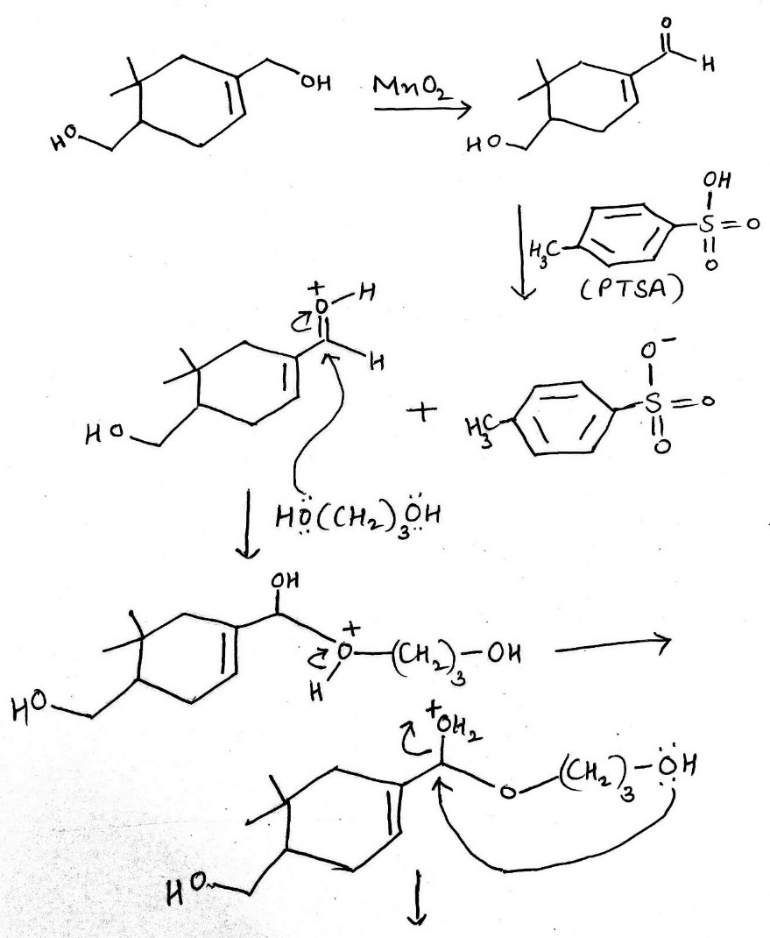
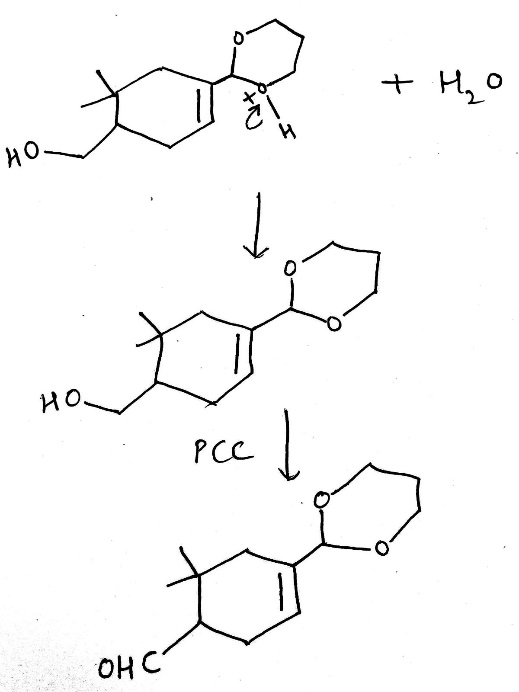
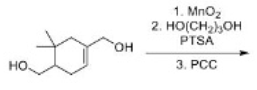
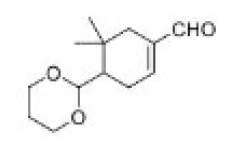
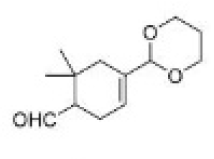
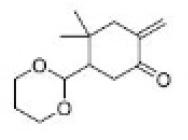
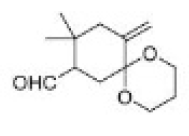
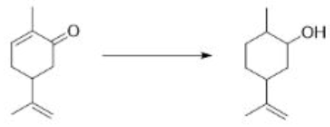
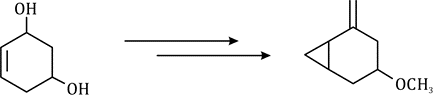
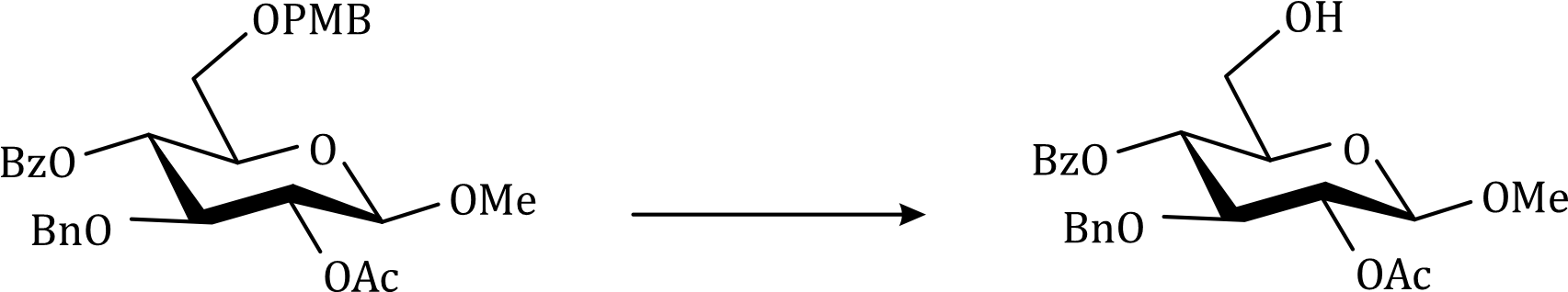
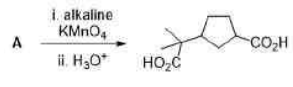
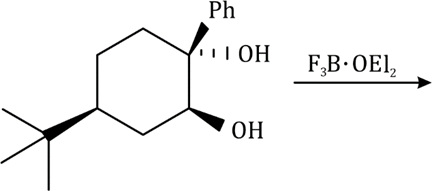
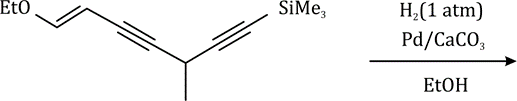Correct option is B
A useful, mild reagent for the oxidation of primary and secondary alcohols to carbonyl compounds is manganese dioxide. This reagent has found most use as a highly specific oxidant for allylic and benzylic hydroxy groups, and reaction takes place under mild conditions (room temperature) in a neutral solvent (e.g. water, petroleum, acetone, DMF, CH2Cl2 or CHCl3). It avoids some of the problems of chromium reagents, which may promote epoxidation of the allylic alcohol or isomerization ( Z to E geometry) of the double bond. For maximum activity it is best to prepare the manganese dioxide immediately prior to use. The best method appears to be by reaction of manganese (II) sulfate with potassium permanganate in alkaline solution; the hydrated manganese dioxide obtained is highly active.
Pyridinium chlorochromate (PCC) is a yellow-orange salt with the formula [C5H5NH]+ [CrO3Cl]- . It is a reagent in organic synthesis used primarily for oxidation of alcohols to form carbonyls. In particular, it has proven to be highly effective in oxidizing primary and secondary alcohols to aldehydes and ketones, respectively.