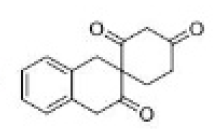
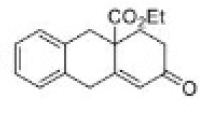
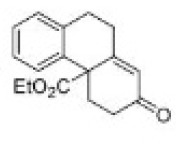
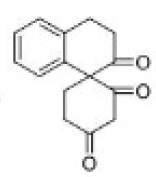
Correct option is C
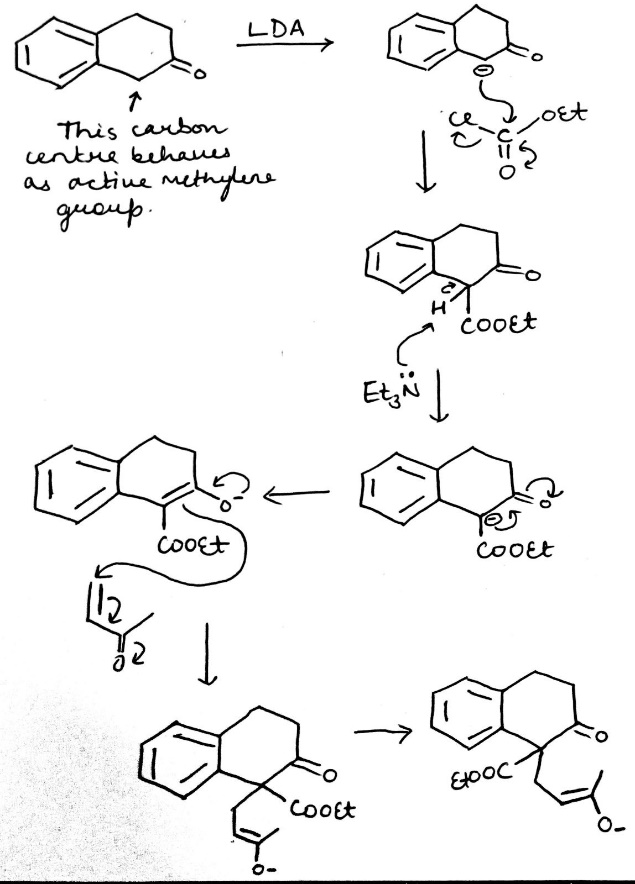
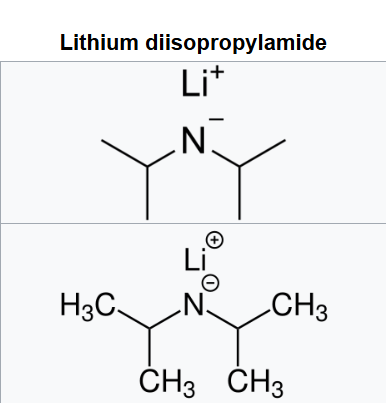
Lithium diisopropylamide (commonly abbreviated LDA) is a chemical compound with the molecular formula LiN(CH(CH3)2)2 . It is used as a strong base and has been widely utilized due to its good solubility in non-polar organic solvents and non-nucleophilic nature.
Lithium enolates are usually made at low temperature in THF with a hindered lithium amide base (often LDA) and are stable under those conditions because of the strong O–Li bond. The formation of the enolate begins with Li–O bond formation before the removal of the proton from the a position by the basic nitrogen atom.
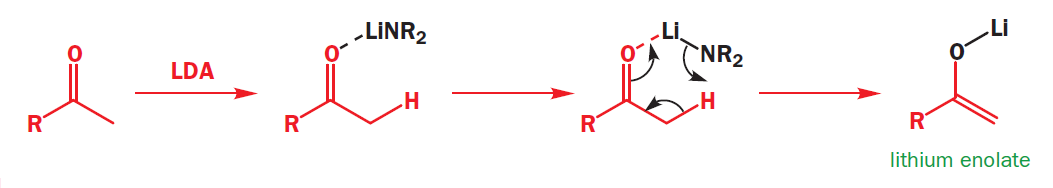
LDA is too hindered to attack C=O, so it attacks C–H instead. And, if there is a choice of C–H bonds, it will attack the least hindered possible. It will also prefer to attack more acidic C–H bonds, and C–H bonds on less substituted carbons are indeed more acidic.
The central carbon in 1,3-dicarbonyl compound is known as an activated methylene group. This is because, owing to the structure, the carbon is especially acidic and can easily be deprotonated to form a methylene group.
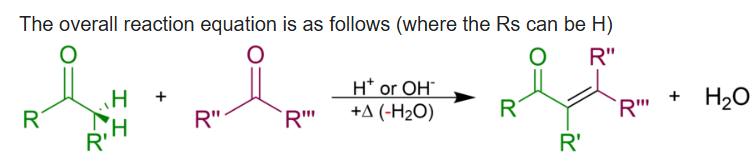
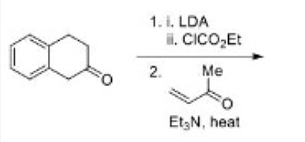
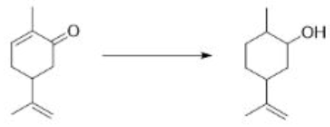
The Robinson annulation is a chemical reaction used in organic chemistry for ring formation. It was discovered by Robert Robinson as a method to create a six membered ring by forming three new carbon–carbon bonds. The method uses a ketone and a methyl vinyl ketone to form an α,β-unsaturated ketone in a cyclohexane ring by a Michael addition followed by an aldol condensation. This procedure is one of the key methods to form fused ring systems.
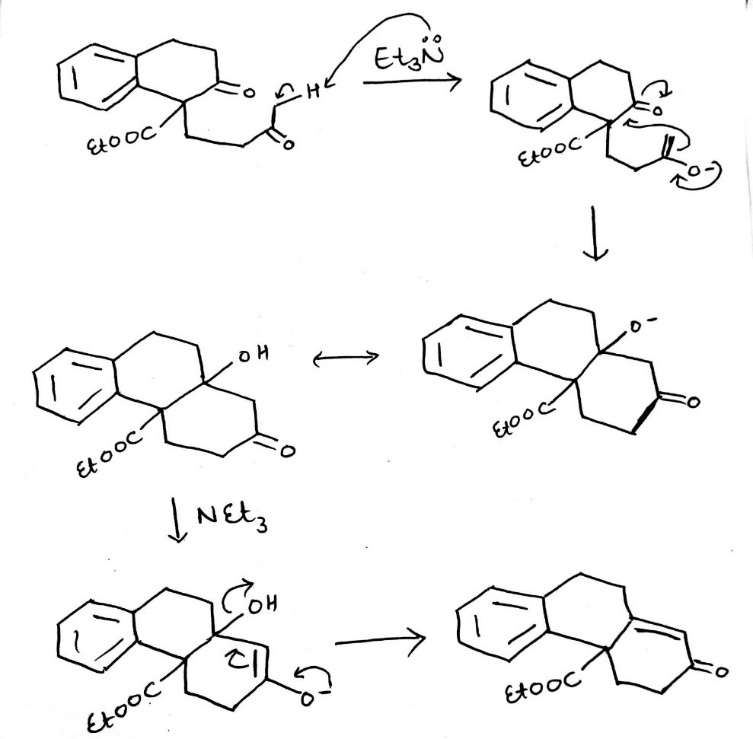
The mechanism combines two important reactions. The first stage is the formation of the stable enolate and the conjugate addition to the enone. The second stage is the formation of a new enolate on the other side of the ketone from the first. Note that the original enolate, the intermediate in the conjugate addition, can cyclize to give only an unstable four-membered ring so this cyclization would be reversible. The next intermediate, the aldol product, is often isolated from Robinson annelations. The final stage is dehydration of the aldol and an E1cB reaction that involves the carbonyl group as in a standard aldol reaction. Another enolate must form in the same position as the last.
Additional Knowledge
In organic chemistry, the Michael reaction or Michael 1,4 addition is a reaction between a Michael donor (an enolate or other nucleophile) and a Michael acceptor (usually an α,β-unsaturated carbonyl) to produce a Michael adduct by creating a carbon-carbon bond at the acceptor's β-carbon.
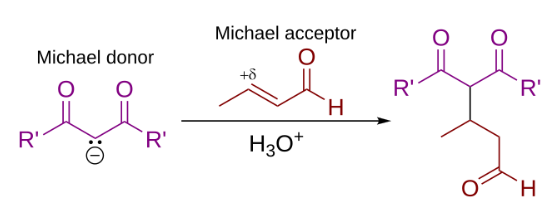
An aldol condensation is a condensation reaction in organic chemistry in which two carbonyl moieties (of aldehydes or ketones) react to form a β-hydroxyaldehyde or β-hydroxyketone (an aldol reaction), and this is then followed by dehydration to give a conjugated enone.