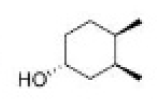
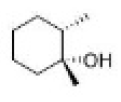
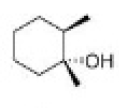
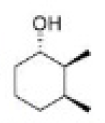
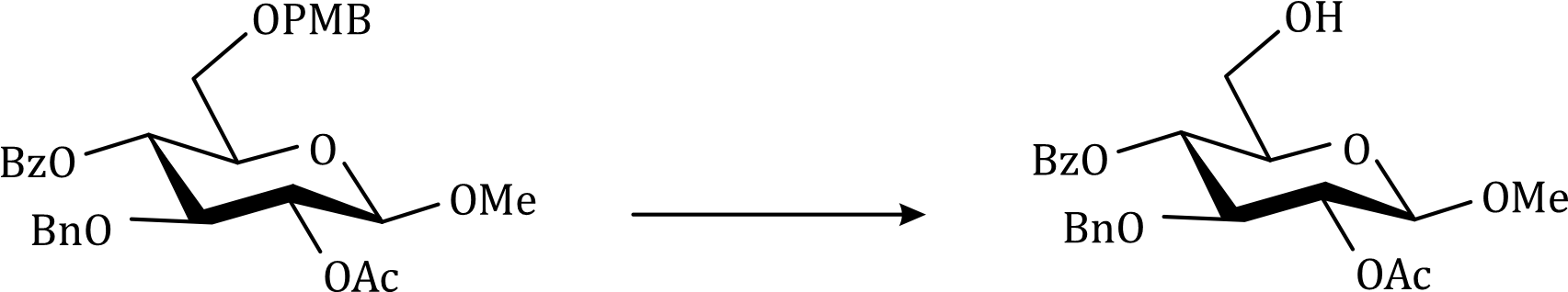
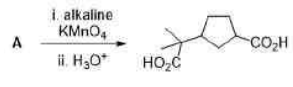
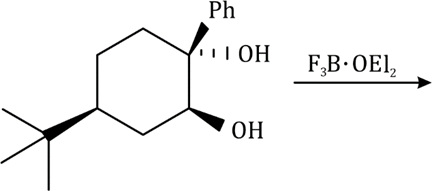
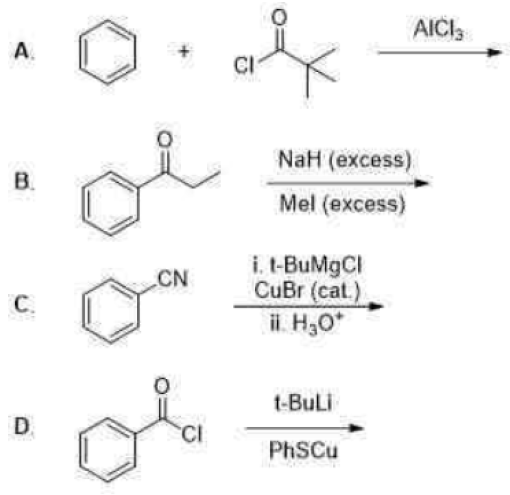
Correct option is C
Step 1:
Group 1 metals, such as sodium or lithium, readily give up their single outer-shell electron as they dissolve in solvents such as liquid ammonia or ethanol. Electrons are the simplest reducing agents, and they will reduce carbonyl compounds, alkynes, or aromatic rings—in fact any functional group with a low-energy π* orbital into which the electron can go.
The dissolving metal reduction of aromatic rings is known as the Birch reduction.
The first thing to note is that when lithium or sodium dissolve in ammonia they give an intense blue solution. Blue is the colour of solvated electrons: these group 1 metals ionize to give Li+ or Na+ and e-(NH3)n- the gaps between the ammonia molecules are just the right size for an electron. With time, the blue colour fades, as the electrons reduce the ammonia to (NH2)- and hydrogen gas.
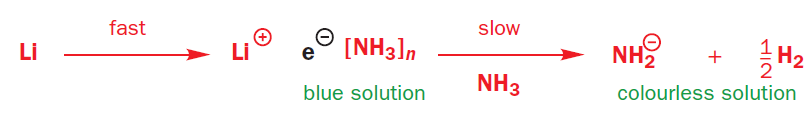
Birch reductions use those blue solutions, with their solvated electrons, as reducing agents. The reduction of NH3 to (NH2)- and H2 is quite slow, and a better electron acceptor will get reduced in preference. The electrons go into benzene’s lowest lying antibonding orbital (its LUMO).
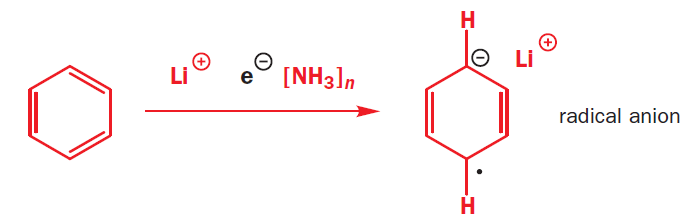
The radical anion is very basic, and it picks up a proton from the ethanol that is in the reaction mixture. The molecule is now no longer anionic, but it is still a radical. It can pick up another electron, which pairs with the radical to give an anion, which is quenched again by the proton source (ethanol).
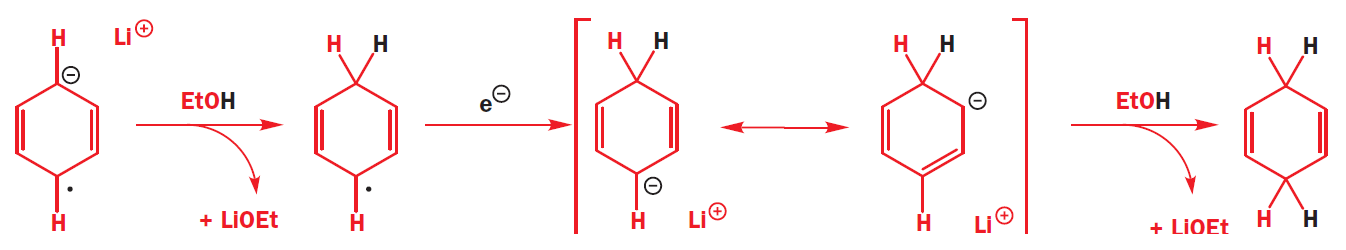
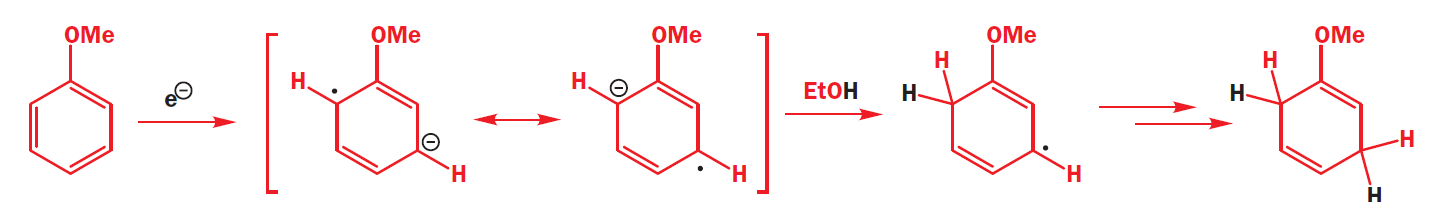
Further questions of regioselectivity arise when there are substituents around the aromatic ring.
Electron-withdrawing groups promote ipso, para reduction while electron-donating groups promote ortho, meta reduction. The explanation must lie in the distribution of electron density in the intermediate radical anions. Electron-donating groups stabilize ortho and meta electron density.
Step 2:
One of the most studied catalytic systems is the Rh(I) complex [RhCl(PPh3)3 ], which is often referred to as Wilkinson’s catalyst. This useful catalyst hydrogenates a wide variety of alkenes and alkynes at pressures of hydrogen close to 1 atm or less at room temperature. The dominant cycle for the hydrogenation of terminal alkenes by Wilkinson’s catalyst is shown
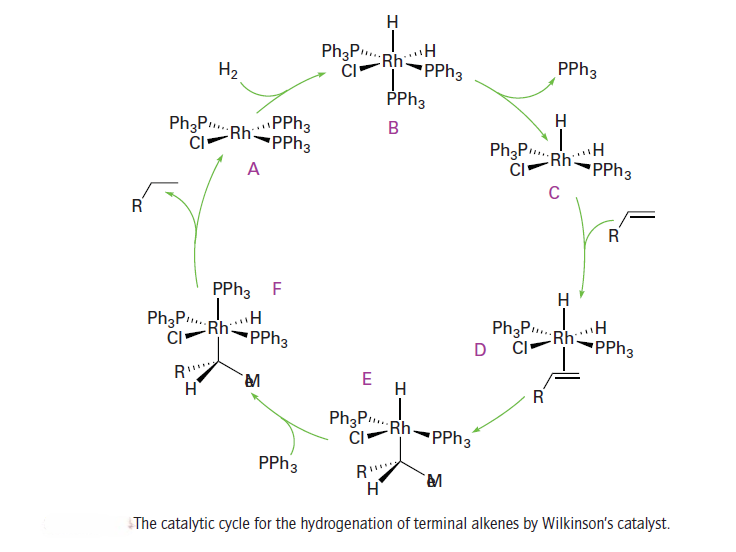
It involves the oxidative addition of H2 to the 16-electron complex [RhCl (PPh3)3] (A), to form the 18-electron dihydrido complex (B). The dissociation of a phosphine ligand from (B) results in the formation of the coordinatively unsaturated complex (C), which then forms the alkene complex (D). Hydrogen transfer from the Rh atom in (D) to the coordinated alkene yields a transient 16-electron alkyl complex (E). This complex takes on a phosphine ligand to produce (F), and hydrogen migration to carbon results in the reductive elimination of the alkane and the reformation of (A), which is set to repeat the cycle. A parallel but slower cycle (which is not shown) is known in which the order of H2 and alkene addition is reversed. Another cycle is known, based around the 14-electron intermediate [RhCl(PPh3)2]. Even though there is very little of this species present, it reacts much faster with hydrogen than [RhCl(PPh3)3] and makes a significant contribution to the catalytic cycle. In this cycle, (E) would eliminate alkane directly, regenerating [RhCl(PPh3)2], which rapidly adds H2 to give (C).
Wilkinson’s catalyst is highly sensitive to the nature of the phosphine ligand and the alkene substrate. The alkene must be just the right size: highly hindered alkenes or the sterically unencumbered ethene are not hydrogenated by the catalyst, presumably because the sterically crowded alkenes do not coordinate and ethene forms a strong complex that does not react further.
Step 3:
Hydroboration–oxidation reaction is a two-step hydration reaction that converts an alkene into an alcohol. The process results in the syn addition of a hydrogen and a hydroxyl group where the double bond had been. Hydroboration–oxidation is an anti-Markovnikov reaction, with the hydroxyl group attaching to the less-substituted carbon.
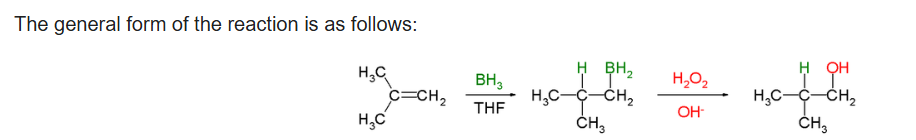
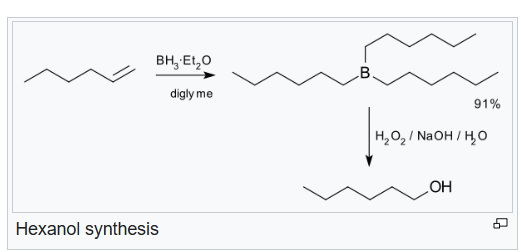
Hydroboration step: In the first step, borane (BH3) adds to the double bond, transferring one of the hydrogen atoms to the carbon adjacent to the one that becomes bonded to the boron. This hydroboration is repeated two additional times, successively reacting each B–H bond so that three alkenes add to each BH3 . The resulting trialkylborane is treated with hydrogen peroxide in the second step. This process replaces the B-C bonds with HO-C bonds. The boron reagent is converted to boric acid.
Oxidation step: In the second step of the reaction sequence, the nucleophilic hydroperoxide anion attacks the boron atom. Alkyl migration to oxygen gives the alkyl borane with retention of stereochemistry (in reality, the reaction occurs via the trialkyl borate B(OR)3 , rather than the monoalkyl borinic ester BH2 OR).
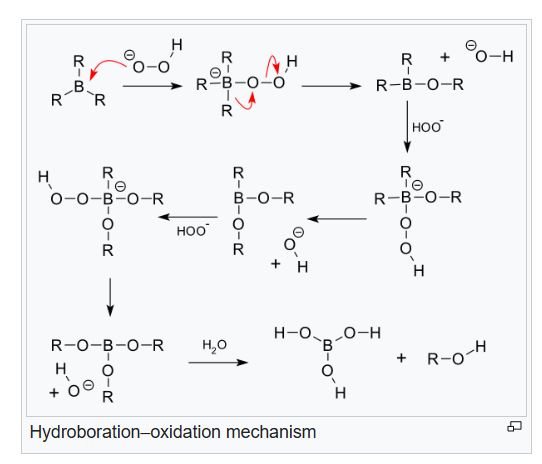
The 'H' atom in the reaction comes from B2H6 , the 'O' atom comes from hydrogen peroxide (H2O2) whereas the O attached 'H' atom comes from the solvent (refer mechanism).