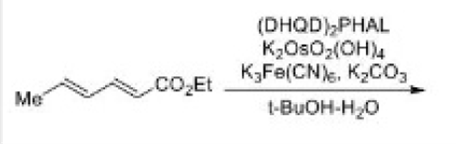
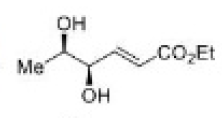
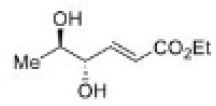
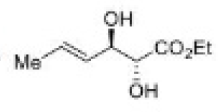
Correct option is A
The dihydroxylation of alkenes provides 1,2-diol products, present in a great many natural products and biologically active molecules. The transformation has, therefore, received considerable interest and methods are now well developed for catalytic, racemic and asymmetric dihydroxylation. The most common of these is syn (or cis) dihydroxylation, in which the two hydroxy groups are added to the same side of the double bond.
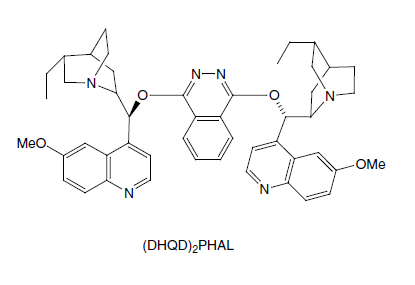
Asymmetric dihydroxylation has been developed into an extremely efficient and selective process for a wide variety of substituted alkenes. Chiral amine ligands provide the required rate enhancement and asymmetric induction, by coordinating to the osmium atom. The most popular ligands are based on the naturally occurring cinchona alkaloids dihydroquinine (DHQ) and dihydroquinidine (DHQD). In particular, the ligands (DHQ)2PHAL and (DHQD)2PHAL, in which two of the alkaloids are connected to a phthalazine ring, have found widespread use.
The dihydroxylation reaction can be carried out with osmium tetroxide as a catalyst (typically added in the lower oxidation state as the solid [K2OsO4·2H2O]), and the favoured co-oxidant is [K3Fe(CN)6].
Due to the high oxidation state of Os (VI), electron rich alkene will convert into a syn diol.