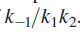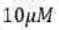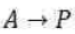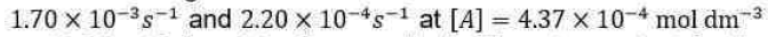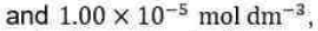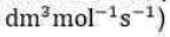Correct option is B
The Lindemann mechanism for unimolecular reactions provides an elegant example of the relationship between kinetics and reaction mechanisms. This mechanism was developed to describe the observed concentration dependence in unimolecular dissociation reactions of the form
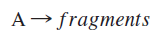
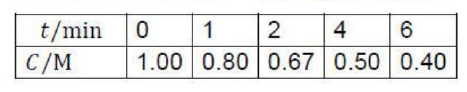
In this reaction, a reactant molecule undergoes decomposition when the energy content of one or more vibrational modes is sufficient for decomposition to occur. The question is “how does the reactant acquire sufficient energy to undergo decomposition?” One possibility is that the reactant acquires sufficient energy to react through a bimolecular collision. Experimentally, however, the rate of decomposition demonstrates only first order behavior at high reactant concentrations, and not second order as expected for a single-step bimolecular mechanism. Frederick Lindemann proposed another mechanism to explain the order dependence of the reaction with respect to reactant concentration.
The Lindemann mechanism involves two steps. First, reactants acquire sufficient energy to undergo reaction through a bimolecular collision:
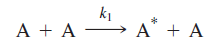
In this reaction, A* is the “activated” reactant that has received sufficient energy to undergo decomposition. The collisional partner of the activated reactant molecule leaves the collision with insufficient energy to decompose. In the second step of the Lindemann mechanism, the activated reactant undergoes one of two reactions: collision resulting in deactivation or decomposition resulting in product formation:
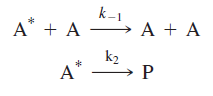
The separation of the reaction into two steps is the key conceptual contribution of the Lindemann mechanism. Specifically, the mechanism implies that a separation in timescale exists between activation and deactivation product formation.
The rate of product production is written as
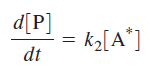
Because A* is an intermediate species, the relationship between [A*] and [A] is obtained by writing the differential rate expression for [A*] and applying the steady-state approximation:
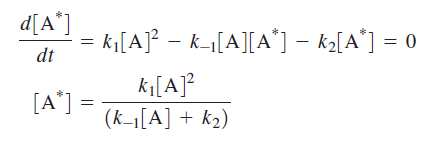
The final differential rate expression for [P]:
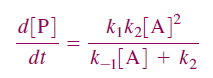
Above equation is the central result of the Lindemann mechanism. It states that the observed order dependence on [A] depends on the relative magnitude of
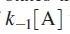
versus k2 . At high reactant concentrations,
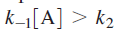
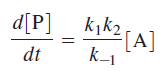
Above equation demonstrates that at high reactant concentrations or pressures (recall that
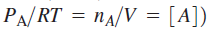
the rate of product formation will be first order in [A], consistent with experiment. Mechanistically, at high pressures activated molecules will be produced faster than decomposition occurs such that the rate of decomposition is the rate-limiting step in product formation. At low reactant concentrations
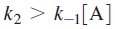
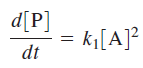
Above equation demonstrates that at low pressures the formation of activated complex becomes the rate-limiting step in the reaction and the rate of product formation is second order in [A].


The Lindemann mechanism provides a detailed prediction of how the rate constant for a unimolecular reaction will vary with pressure or concentration.
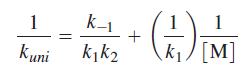
Above equation predicts that a plot of
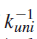
versus
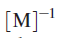
should yield a straight line with
slope
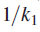
and y intercept of