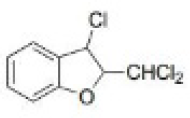
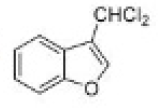
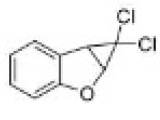
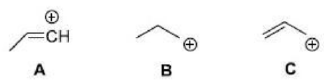
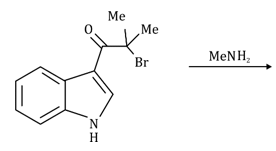
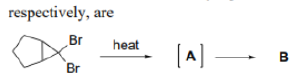
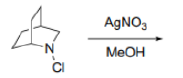
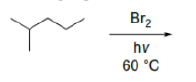
Correct option is D
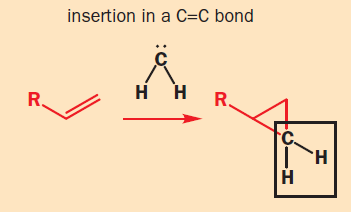
Carbenes are neutral species containing a carbon atom with only six valence electrons. Carbenes have six electrons: two in each bond and two nonbonding electrons, which are often represented as :CR2.
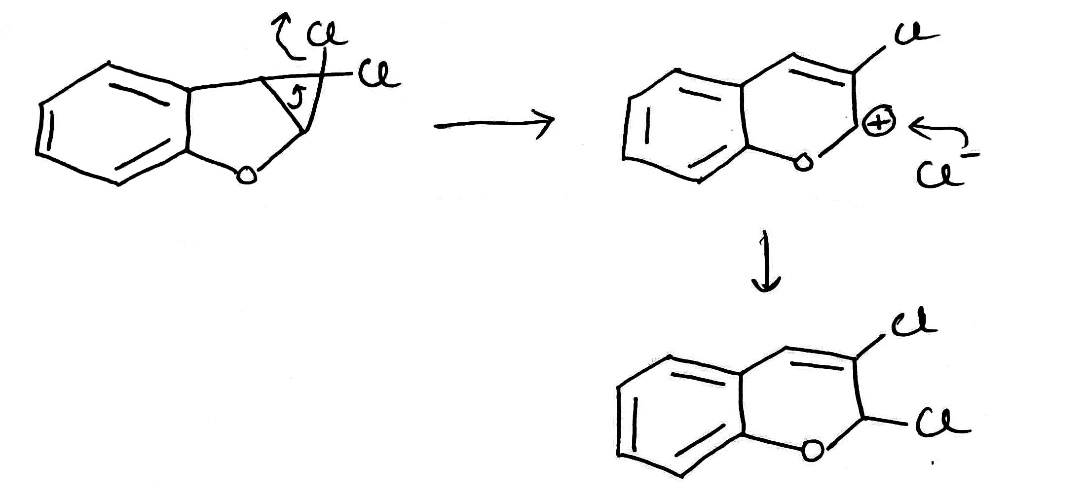
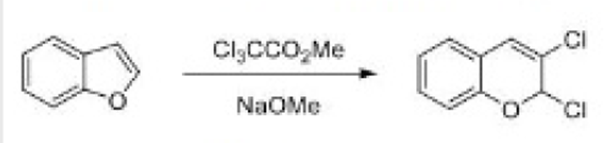
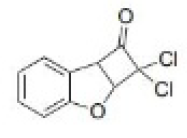
Carbenes are usually formed from precursors by the loss of small, stable molecules.
Decarboxylation of sodium trichloroacetate happens at about 80°C in solution.
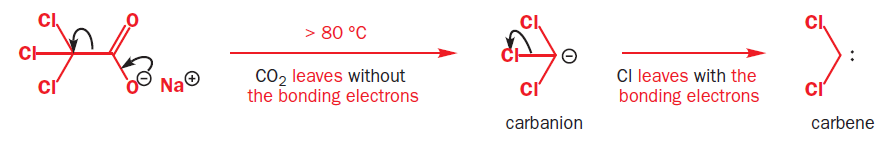
Carbenes are extremely reactive species. They are trapped by alcohols to make ethers, but more importantly they will react with alkenes to make cyclopropanes, and they will also insert into C–H bonds.