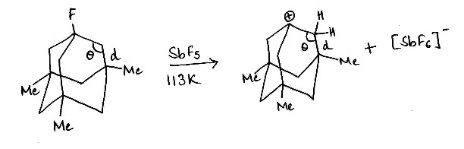
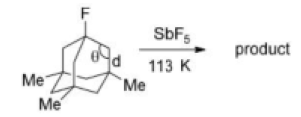
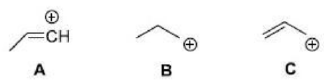
Correct option is A
Antimony pentafluoride is the inorganic compound with the formula SbF5.SbF5 is a strong Lewis acid, exceptionally so toward sources of F- to give the very stable anion [SbF6]-,called hexafluoroantimonate. It is very stable because the negative charge is distributed on six fluorine atoms. The fluorine atoms can “pump” electron density from the antimony atom because the latter's electronegativity is low. It is the conjugate base of the superacid fluoroantimonic acid. Fluoroantimonic acid is a mixture of hydrogen fluoride and antimony penta fluoride, containing various cations and anions (the simplest being [H2F]+[SbF6]-).
Carbon atom containing the fluorine atom is tetrahedral (bond angle ≈ 109.5°). A carbocation will be formed due to a large flexible system. Because of the presence of angle strain in the carbocation intermediate, hyperconjugation will not be possible. Bond length will not reduce and the bond angle will not increase.
Information Booster
Hyperconjugation involves the interaction of the electrons in a sigma (σ) orbital (e.g. C–H or C–C) with an adjacent unpopulated non-bonding p or antibonding σ* or π* orbitals to give a pair of extended molecular orbitals. Hyperconjugation is suggested as a key factor in shortening of sigma bonds (σ bonds).
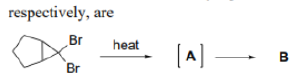
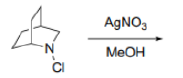
In organic chemistry, an anti-Bredt molecule is a bridged molecule with a double bond at the bridgehead. Bredt's rule is the empirical observation that such molecules only form in large ring systems. Bredt's rule results from geometric strain: a double bond at a bridgehead atom necessarily must be trans in at least one ring. For small rings (fewer than eight atoms), a trans alkene cannot be achieved without substantial ring and angle strain (the p orbitals are improperly aligned for a π bond). Bredt's rule also applies to carbocations because these intermediates also prefer a planar geometry with 120° angles and sp2 hybridization.