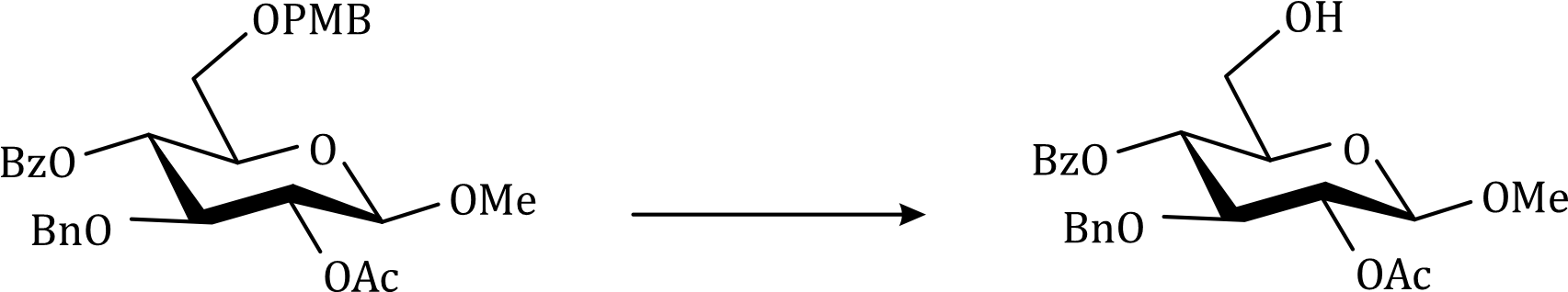
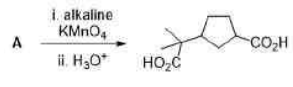
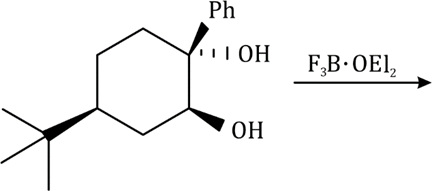
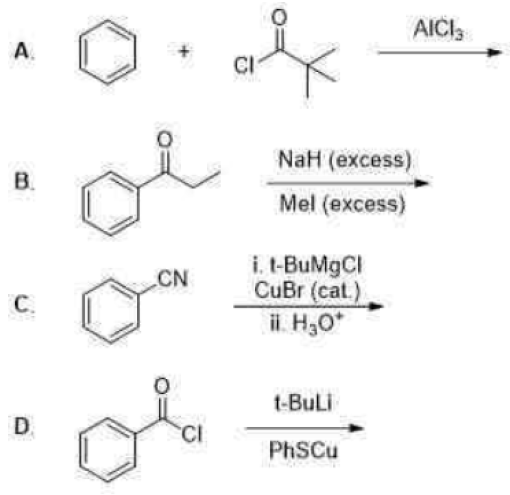
Correct option is C
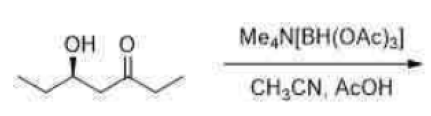
Treatment of β-hydroxy ketones with tetramethylammonium triacetoxyborohydride [Me4NHB(OAc)3] complements the chelation approach by affording the corresponding 1,3-trans diols. Acetic acid-promoted ligand exchange provides an alkoxydiacetoxyborohydride intermediate in which the proximal ketone is stereoselectively reduced by intramolecular hydride transfer. Alkyl substituents in the α-position do not diminish the 1,3-asymmetric induction.
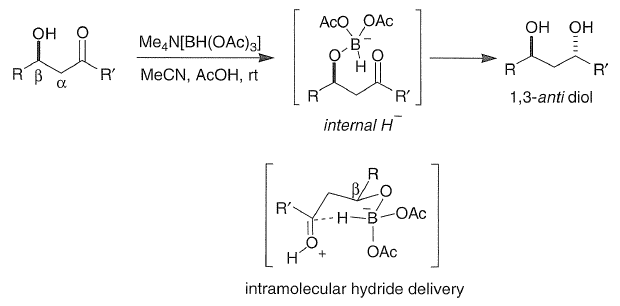
The product obtained is chiral as it does not have a plane of symmetry. Both the OH groups are present in the opposite direction.
Information Booster
The Saksena–Evans reduction is a diastereoselective reduction of β-hydroxy ketones to the corresponding anti-dialcohols, employing the reagent tetramethylammonium triacetoxyborohydride.The reaction is thought to proceed through the 6-membered ring transition state shown below. The intramolecular hydride delivery from the boron reducing agent forces the reduction to proceed from the opposite face of the chelating β-alcohol, thus determining the diastereoselectivity.