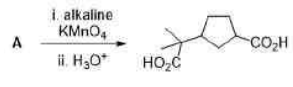
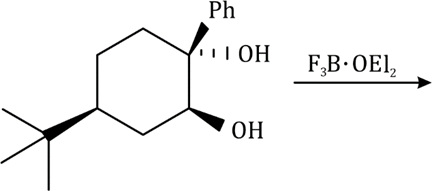
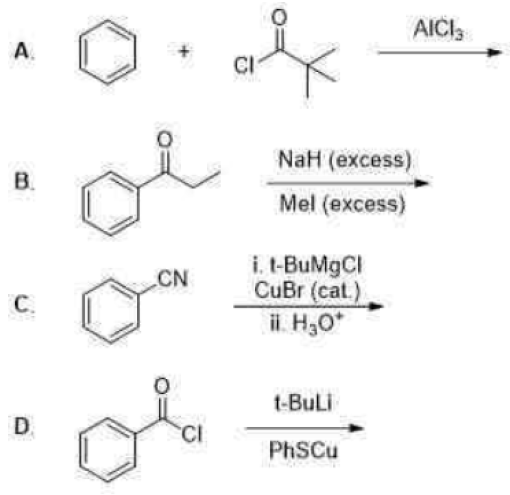
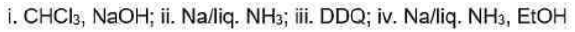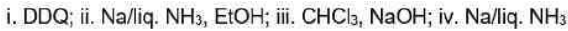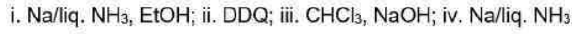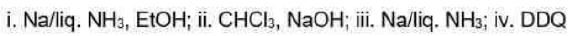Correct option is D
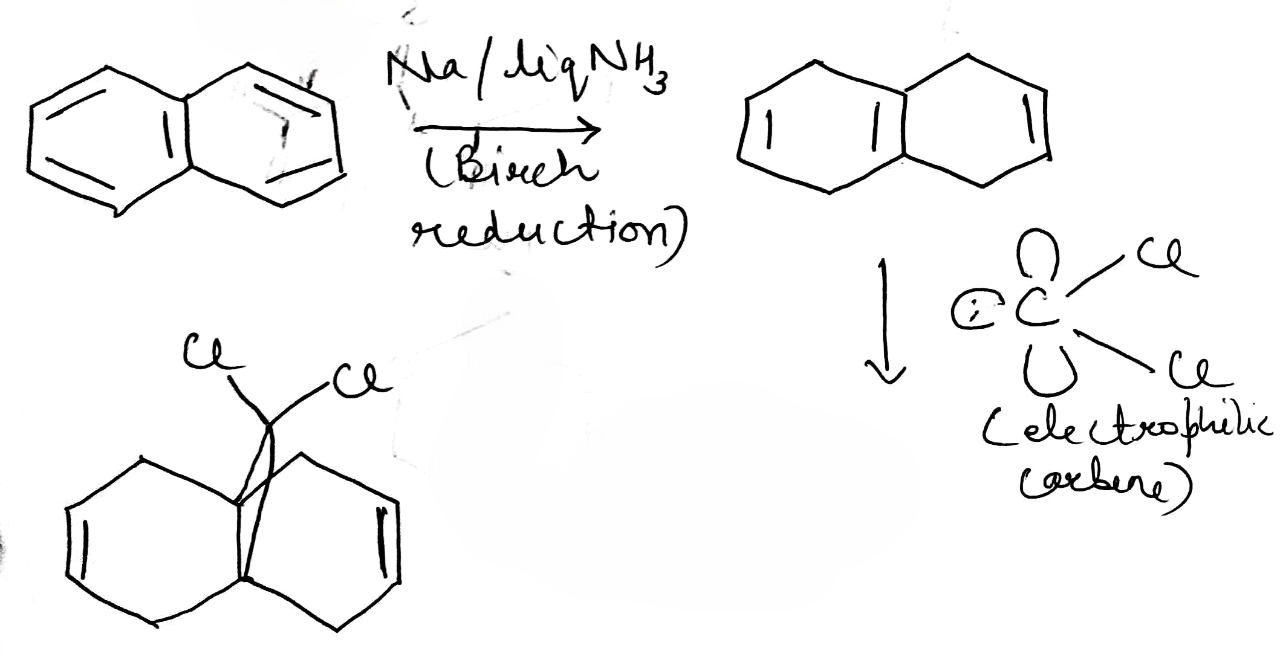
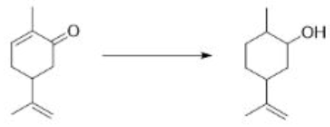
Birch reduction
Group 1 metals, such as sodium or lithium, readily give up their single outer-shell electron as they dissolve in solvents such as liquid ammonia or ethanol. Electrons are the simplest reducing agents, and they will reduce carbonyl compounds, alkynes, or aromatic rings—in fact any functional group with a low-energy π* orbital into which the electron can go.
The dissolving metal reduction of aromatic rings is known as the Birch reduction.
The first thing to note is that when lithium or sodium dissolve in ammonia they give an intense blue solution. Blue is the colour of solvated electrons: these group 1 metals ionize to give Li+ or Na+ and e-(NH3)n-the gaps between the ammonia molecules are just the right size for an electron. With time, the blue colour fades, as the electrons reduce the ammonia to (NH2)- and hydrogen gas.
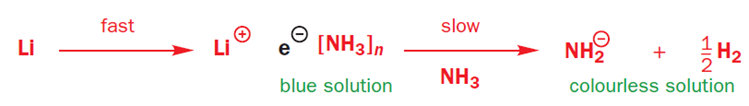
Birch reductions use those blue solutions, with their solvated electrons, as reducing agents. The reduction of NH3 to (NH2)- and H2 is quite slow, and a better electron acceptor will get reduced in preference. The electrons go into benzene’s lowest lying antibonding orbital (its LUMO).
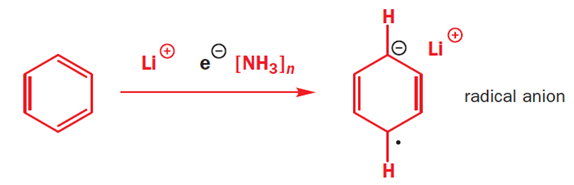
The radical anion is very basic, and it picks up a proton from the ethanol that is in the reaction mixture. The molecule is now no longer anionic, but it is still a radical. It can pick up another electron, which pairs with the radical to give an anion, which is quenched again by the proton source (ethanol).
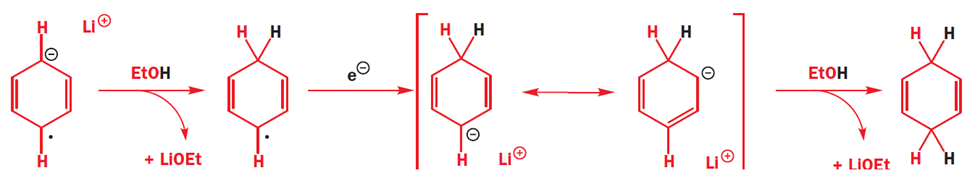
Generation of carbene
One of the best known a elimination reactions occurs when chloroform is treated with base. This is the most important way of making dichlorocarbene, :CCl2, and other dihalocarbenes too, although it must be said that the widespread use of dichlorocarbene in chemistry is due mainly to the ease with which it can be made using this method!
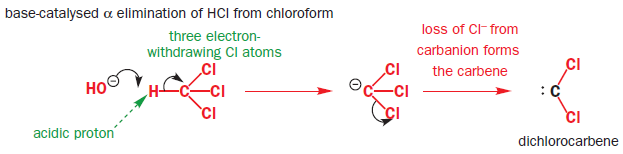
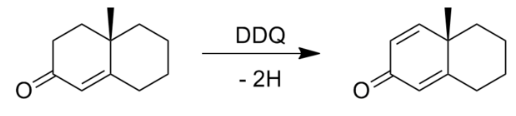
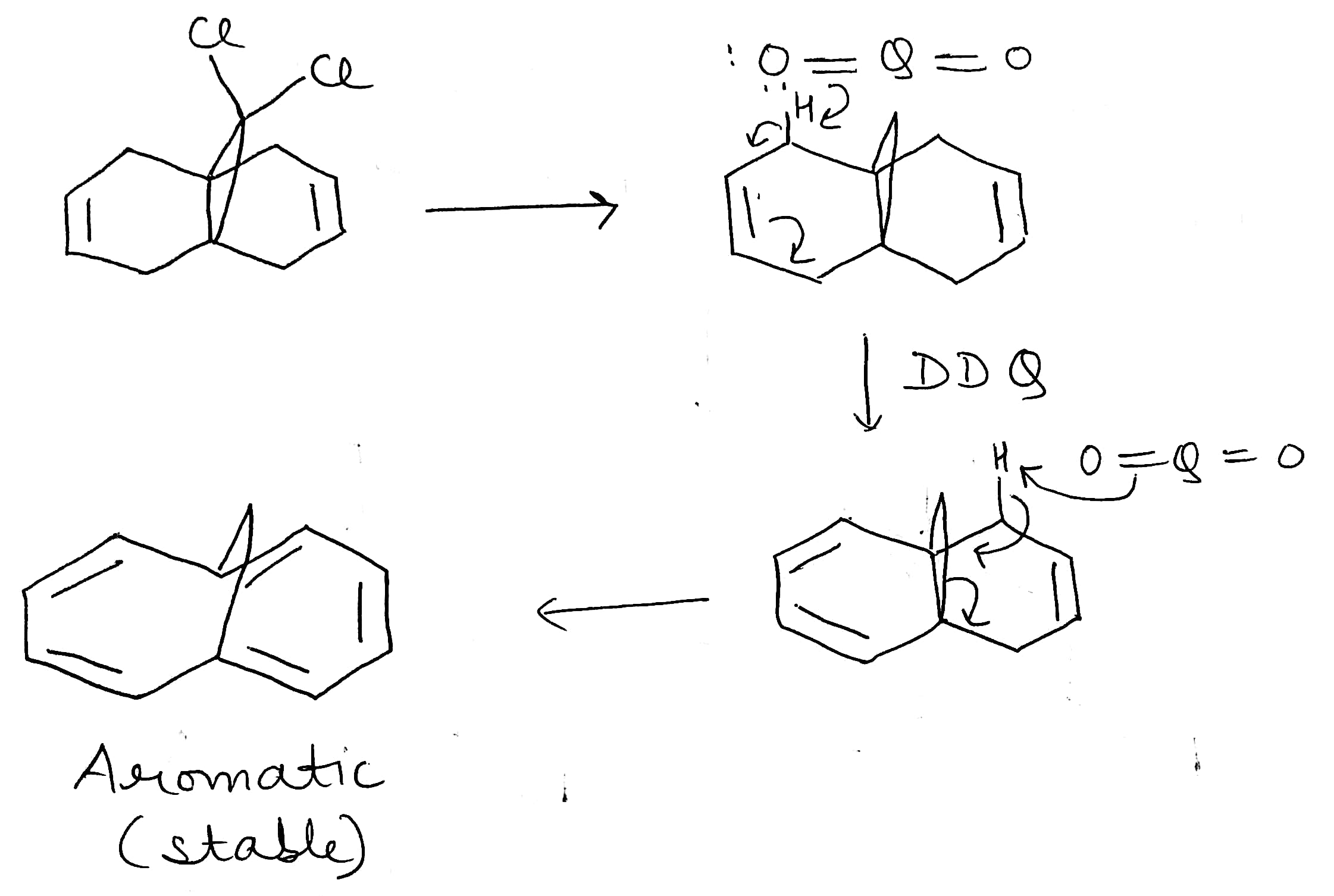
DDQ
2,3-Dichloro-5,6-dicyano-1,4-benzoquinone (or DDQ) is the chemical reagent with formula C6Cl2(CN)2O2.
The reagent removes pairs of H atoms from organic molecules. The stoichiometry of its action is illustrated by the conversion of tetralin to naphthalene: