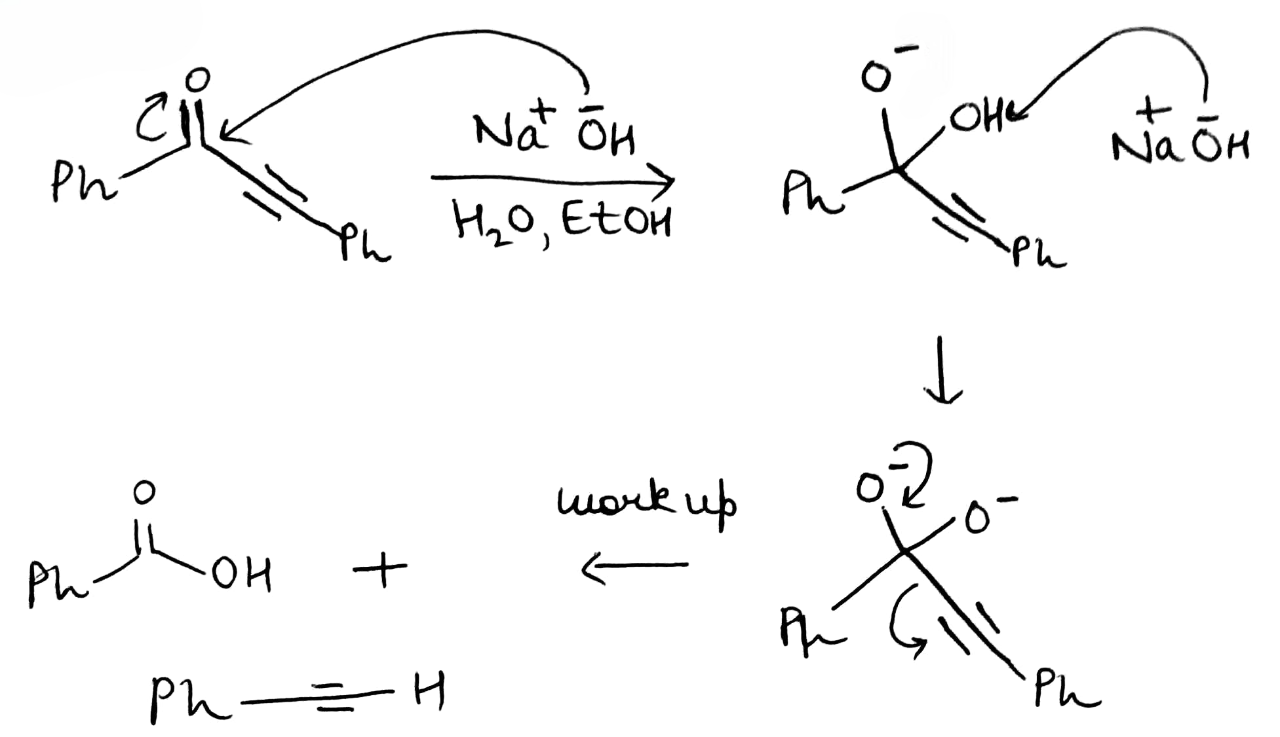
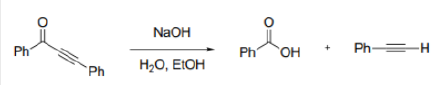
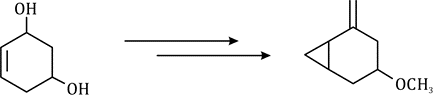
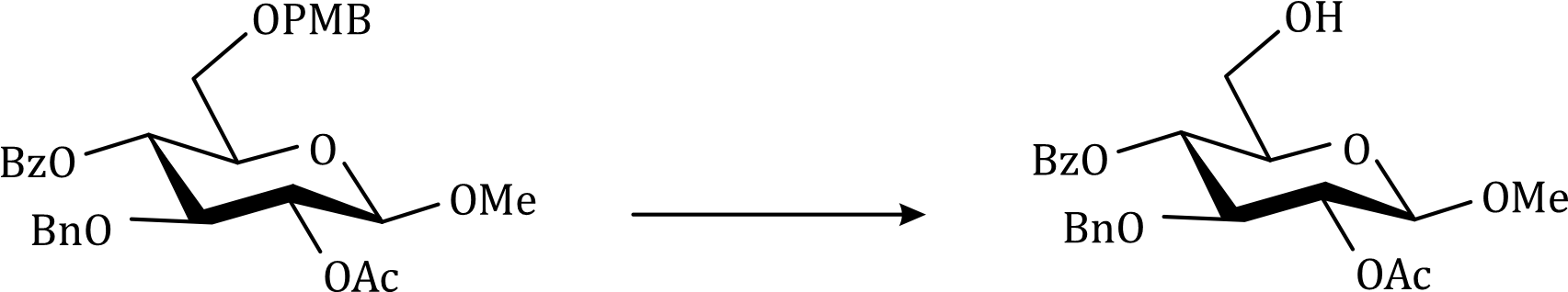
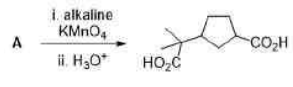
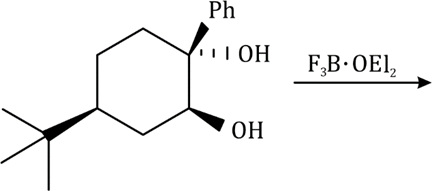
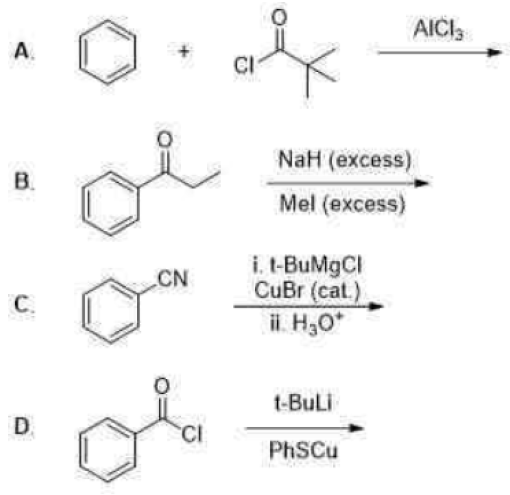
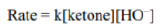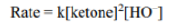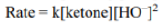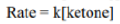Correct option is C
In the given reaction, OH- cannot act as a base due to the absence of an acidic hydrogen in the substrate. Therefore, it acts as a nucleophile and attacks the carbonyl carbon. The second step involves the abstraction of an acidic hydrogen by the OH- ion. The leaving group contains a sp hybridised carbon bearing a negative charge. The sp hybridised carbon having a negative charge is stable to due to its high electronegativity.
The electronegativity of an atom changes depending on the hybridization of the orbital employed in bonding. Electrons in s orbitals are held more tightly than electrons in p orbitals. Hence, a bond to an atom that employs an
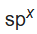
hybrid orbital for bonding will be more heavily polarized to that atom when the hybrid orbital has more s character. That is, when electronegativities are compared for different hybridization schemes of a given element, the order χ(sp3) < χ(sp2) < χ(sp) holds.