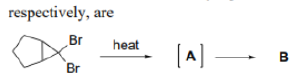
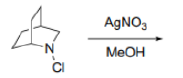
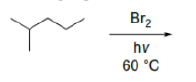
Correct option is B
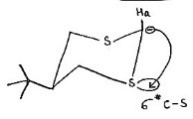
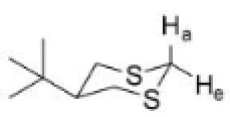
Ha is less acidic than He. The carbanion generated by the removal of Ha will repel more with the lone pairs on sulfur. The carbanion generated by the removal of He will also repel with the lone pairs on sulfur. However, the generated equatorial carbanion is delocalized into the σ* orbital of C-S bond as shown below: