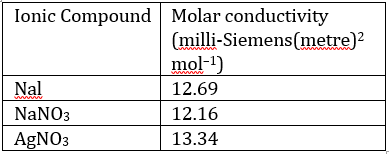
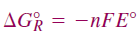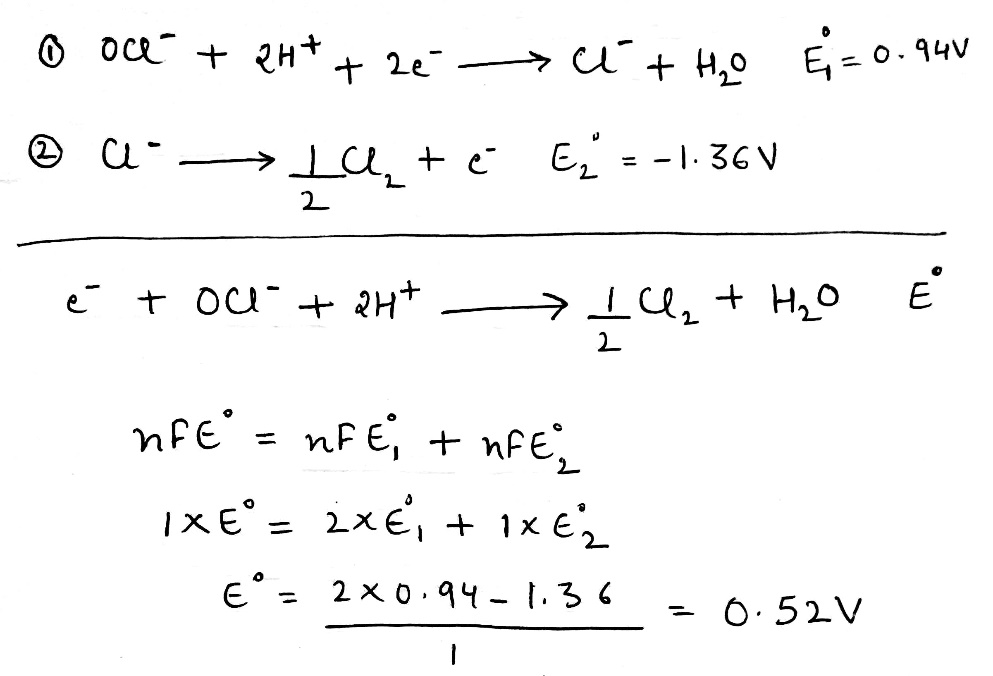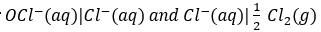Correct option is C
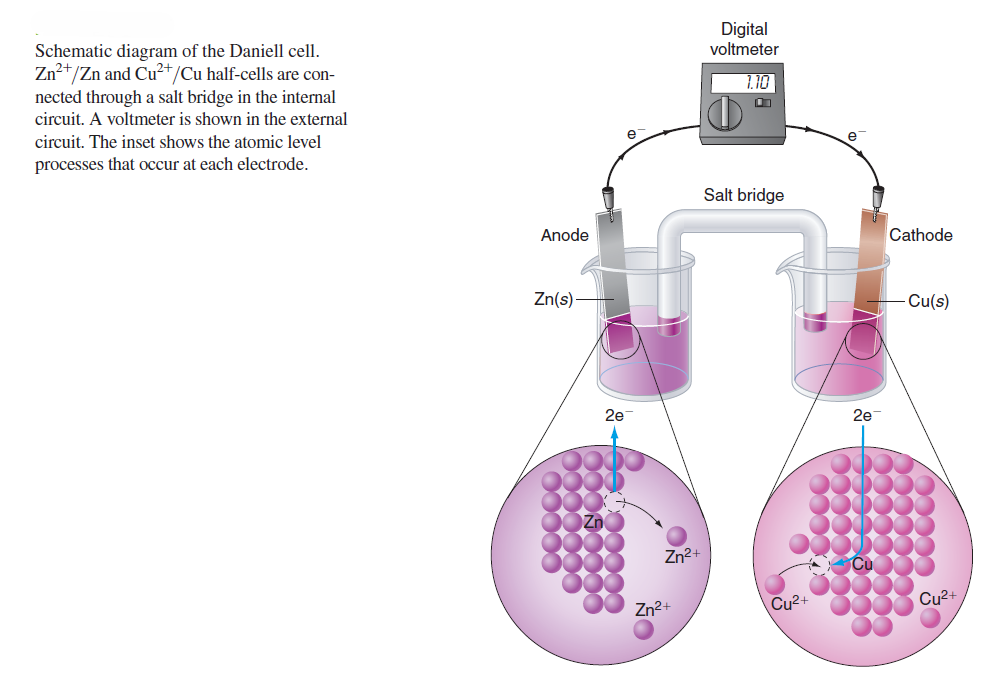
In a Daniel cell, if the half-cells are connected through the external circuit, Zn atoms leave the Zn electrode to form Zn2+ in solution, and Cu2+ ions are deposited as Cu atoms on the Cu electrode. In the external circuit, it is observed that electrons flow through the wires and the resistor in the direction from the Zn electrode to the Cu electrode. These observations are consistent with the following electrochemical reactions:
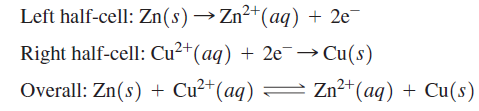
In the left half-cell, Zn is being oxidized to Zn2+, and in the right half-cell, Cu2+ is being reduced to Cu. By convention, the electrode at which oxidation occurs is called the anode, and the electrode at which reduction occurs is called the cathode. Each half-cell in an electrochemical cell must contain a species that can exist in an oxidized and a reduced form. For a general redox reaction, the reactions at the anode and cathode and the overall reaction can be written as follows:
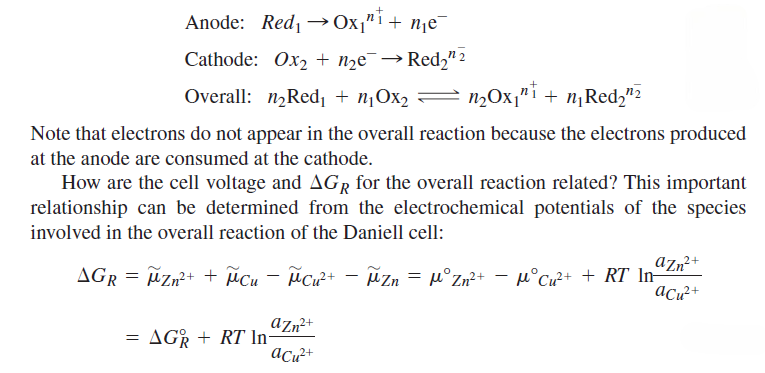
If this reaction is carried out reversibly, the electrical work done is equal to the product of the charge and the potential difference through which the charge is moved. However, the reversible work at constant pressure is also equal to
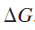
. Therefore, we can write the following equation:


Therefore, if the cell potential is measured under standard conditions,