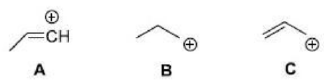
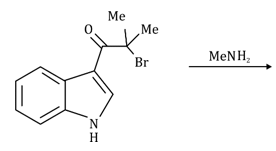
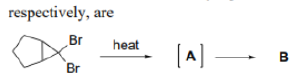
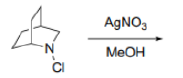
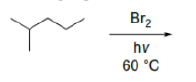
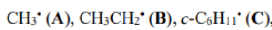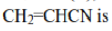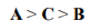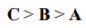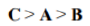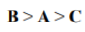Correct option is B
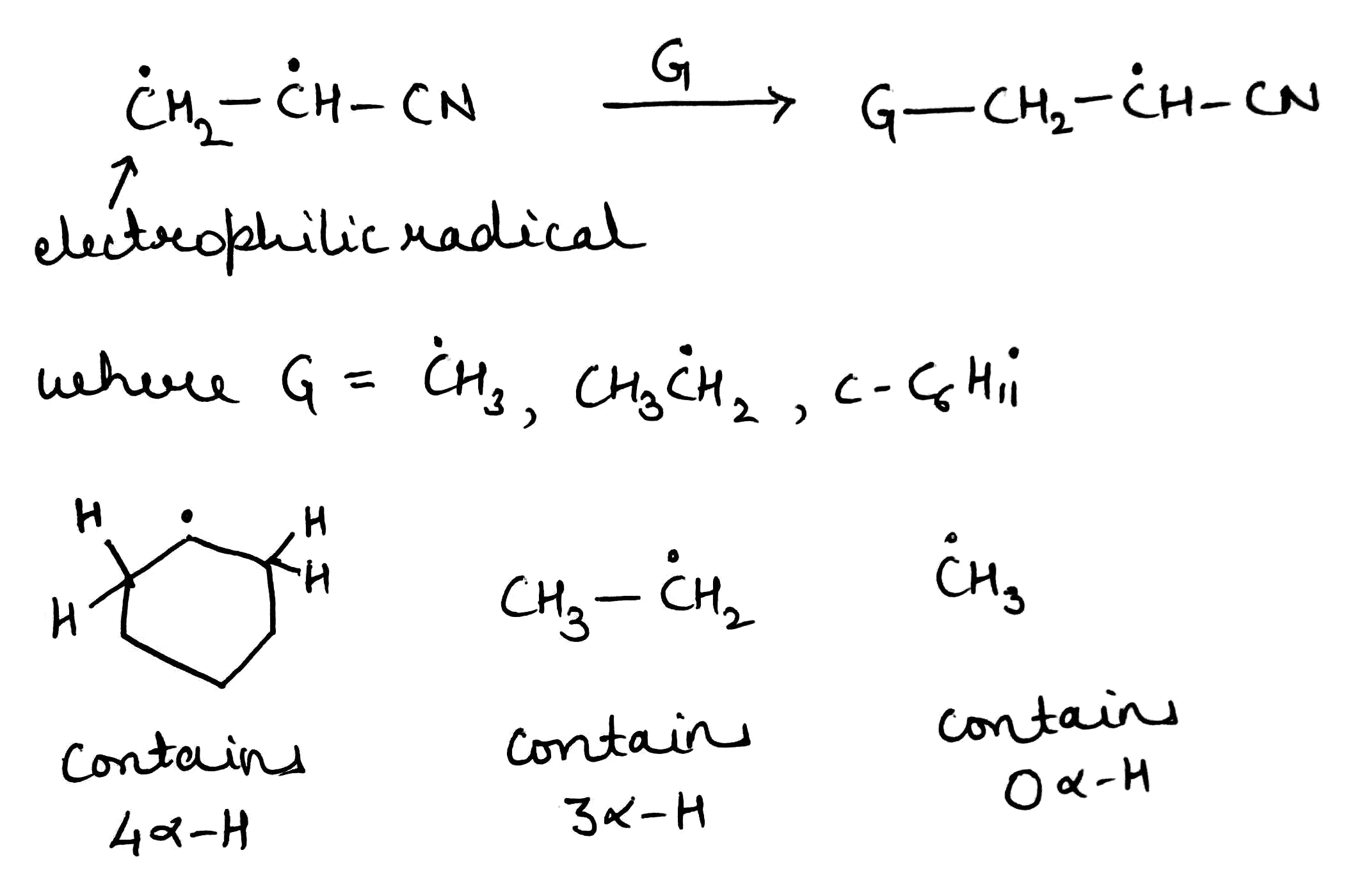
Radicals are stabilized by conjugating, electron-withdrawing, and electron donating groups
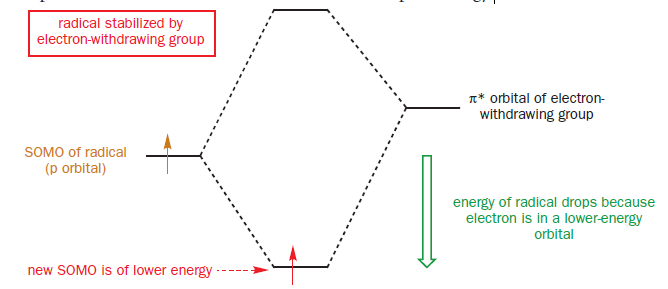
Groups like C=O and CN are electron-withdrawing because they have a low-lying empty п* orbital. By overlapping with the (usually p) orbital containing the radical (the SOMO), two new molecular orbitals are generated. One electron (the one in the old SOMO) is available to fill the two new orbitals. It enters the new SOMO, which is of lower energy than the old one, and the radical experiences stabilization because this electron drops in energy.
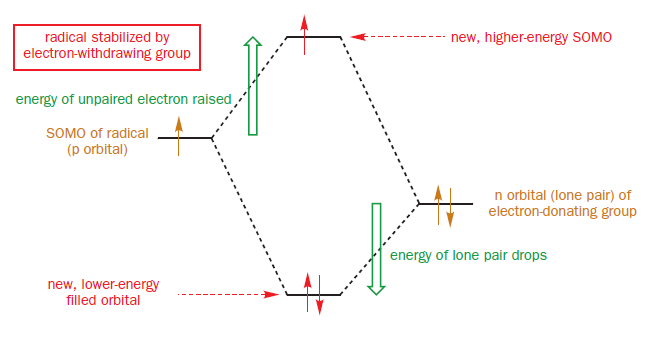
Ether oxygen atoms have relatively high-energy filled n orbitals, their lone pairs. Interacting this with the SOMO again gives two new molecular orbitals. Three electrons are available to fill them. The SOMO is now higher in energy than it was to start with, but the lone pair is lower. Because two electrons have dropped in energy and only one has risen, there is an overall stabilization of the system, even though the new SOMO is of higher energy than the old one.
Hyperconjugation (σ-conjugation or no-bond resonance) refers to the delocalization of electrons with the participation of bonds of primarily σ-character. Usually, hyperconjugation involves the interaction of the electrons in a sigma (σ) orbital (e.g. C–H or C–C) with an adjacent unpopulated non-bonding p or antibonding σ* or π* orbitals to give a pair of extended molecular orbitals.
In radical chemistry, radicals are stabilized by hyperconjugation with adjacent alkyl groups. The donation of sigma (σ) C−H bonds into the partially empty radical orbitals helps to differentiate the stabilities of radicals on tertiary, secondary, and primary carbons. Tertiary carbon radicals have three σ C-H bonds that donate, secondary radicals only two, and primary radicals only one. Therefore, tertiary radicals are the most stable and primary radicals the least stable.