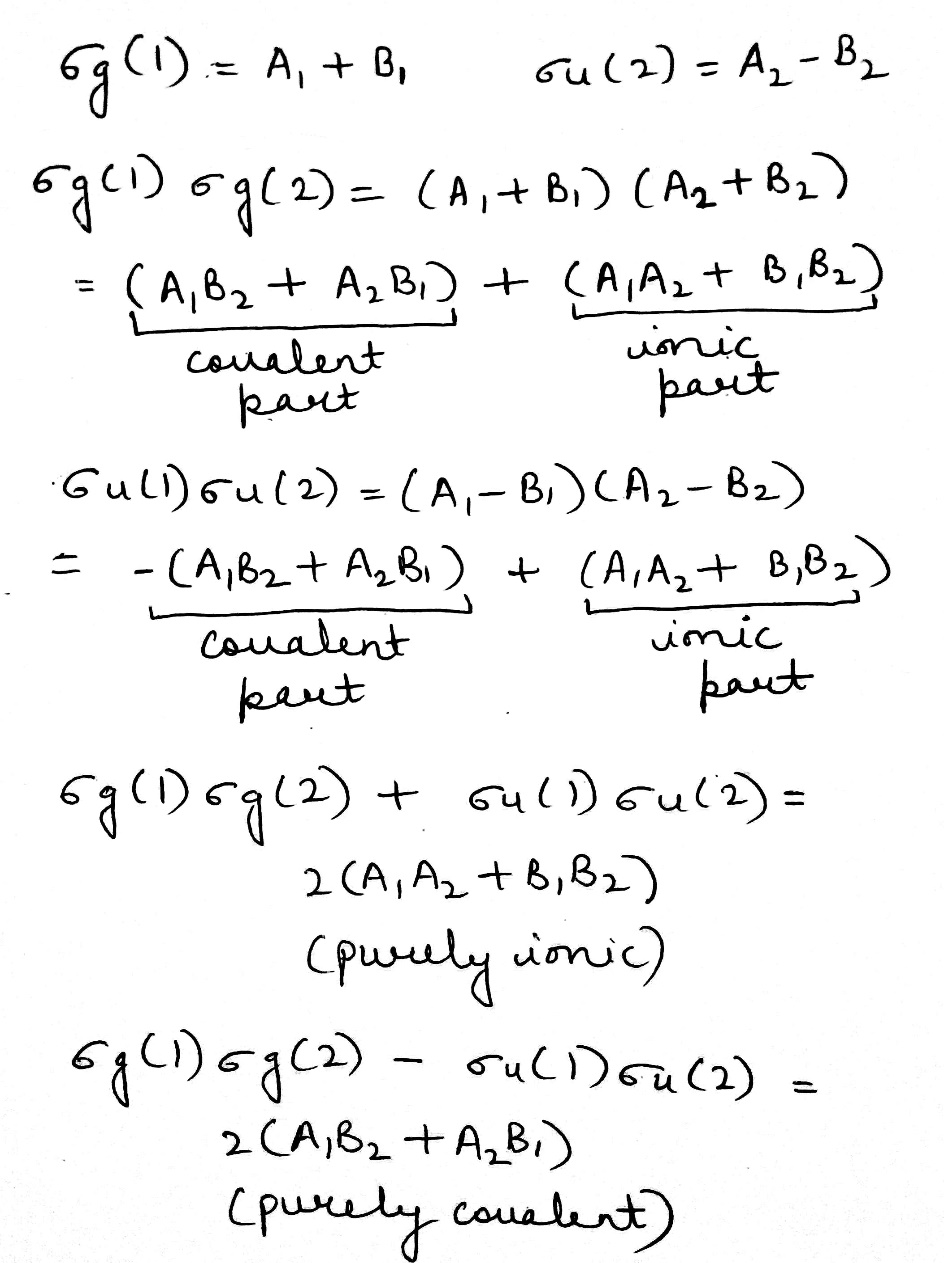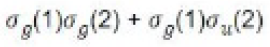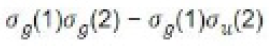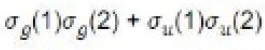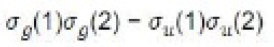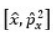Correct option is D
The hydrogen molecule contains two protons and two electrons.
The two electrons will occupy the lowest bonding molecular orbital ψ+ with opposite spins. The total molecular wave function will include both the spatial and spin functions and will be given by




Since the coefficients of all the terms are identical, it follows that MO treatment gives equal weightage to the ionic and covalent structures. It is expected that the MO function will be poor at large internuclear separations since the dissociation products will be an equal mixture of ions and atom. In actual practice we get two hydrogen atoms. On the other hand, for very short internuclear distances one expects that there will also be some chance of finding both the electrons in the same atomic orbital. Thus, MO treatment does take into account this fact, but perhaps gives more weightage to this fact than what actually exists in the molecule.
The MO can be improved by the method of configuration interaction. In this, the wave function is taken as linear combination of functions arising from different electronic configurations. For hydrogen molecule, the following four spatial wave functions are possible:

Those configurations will mix which have the same symmetry with respect to the exchange of the two protons. Remembering that ψ+ and ψ- are symmetrical and antisymmetrical wave functions, respectively, the symmetry nature of each of the above configurations can be worked out as follows