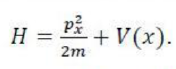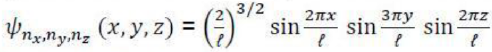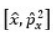Correct option is D
A molecular configuration is a specification of the occupied molecular orbitals in a molecule. A given configuration may have several different states depending on how the electrons are arranged in the valence orbitals); a molecular term symbol labels these states and specifies the total spin and orbital angular momentum of the molecule, along with various other symmetries. The term symbols are of the form:
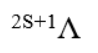

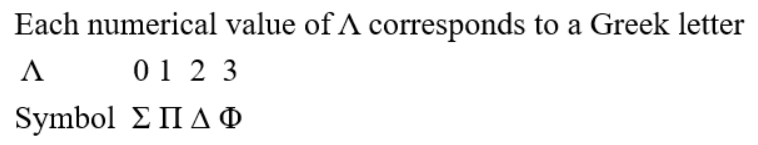
The g/u subscript applies only to molecules with a centre of symmetry. For homonuclear diatomic molecules, there is inversion symmetry. This can be thought of as simple multiplication of functions of even or odd symmetry. Thus, for a pair of electrons in the same σg MO, the product would be (gerade)(gerade), which is (even)(even) = even; thus the symmetry is gerade.
In general: (g)(g) = g; (g)(u) = u; (u)(u) = g
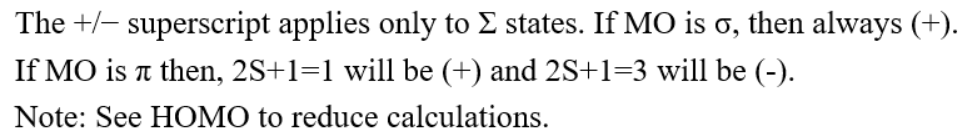
A nitrogen atom has 2+5=7 electrons. Thus, the N2 molecule contains 14 electrons. These are arranged in MOs
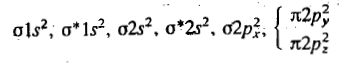
For N2+,
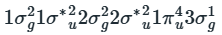
The unpaired electron is present in a sigma orbital (σg).
2S+1=2(1/2)+1=2