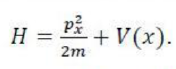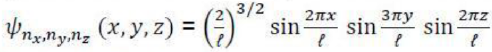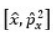Correct option is B
In quantum mechanics, the expectation value is the probabilistic expected value of the result (measurement) of an experiment. It can be thought of as an average of all the possible outcomes of a measurement as weighted by their likelihood, and as such it is not the most probable value of a measurement; indeed the expectation value may have zero probability of occurring (e.g. measurements which can only yield integer values may have a non-integer mean).
Consider an operator A. The expectation value is then in Dirac notation with a normalized state vector.
The normalized ground state wavefunction for one electron system like He+ is: