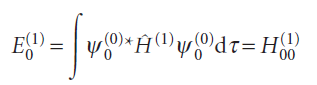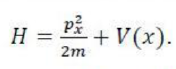Correct option is B
Many problems-and almost all the problems of interest in chemistry-do not have exact solutions. To make progress with these problems we need to develop techniques of approximation. There are two major approaches, variation theory and perturbation theory.
Time-independent perturbation theory
In perturbation theory, we suppose that the Hamiltonian for the problem we are trying to solve,
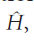
can be expressed as the sum of a simple Hamiltonian,
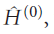
which has known eigenvalues and eigenfunctions, and a contribution,
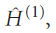
which represents the extent to which the true Hamiltonian differs from the ‘model’ Hamiltonian:
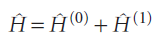
In time-independent perturbation theory, the perturbation is always present and unvarying. For example, it might represent a dip in the potential energy of a particle in a box in some region along the length of the box. In time-independent perturbation theory, we suppose that the true energy of the system differs from the energy of the simple system, and that we can write
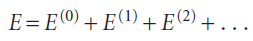

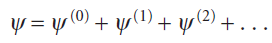
The first-order correction to the energy of the ground state (with the wavefunction ψ0 and energy E0), is