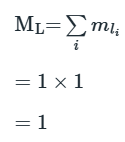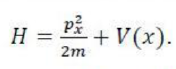Correct option is B
For any system containing more than one electron, the energy of an electron with principal quantum number n depends on the value of l, and this also determines the orbital angular momentum which is given by
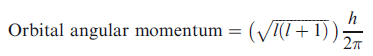
The energy and the orbital angular momentum of a multielectron species are determined by a new quantum number, L, which is related to the values of l for the individual electrons. Since the orbital angular momentum has magnitude and (2l+1) spatial orientations with respect to
the z axis (i.e. the number of values of ml), vectorial summation of individual l values is necessary. The value of ml for any electron denotes the component of its orbital angular momentum, ml (h/2π), along the z axis. Summation of ml values for individual electrons in a multi-electron system therefore gives the resultant orbital magnetic quantum number ML:
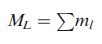
Just as ml may have the (2l+1) values l, (l-1)...0...-(l -1), -l, so ML can have (2L+1) values L, (L-1) ... 0 ...-(L-1), -L. If we can find all possible values of ML for a multielectron species, we can determine the value of L for the system.
As a means of cross-checking, it is useful to know what values of L are possible. The allowed values of L can be determined from l for the individual electrons in the multi-electron system. For two electrons with values of l1 and l2:
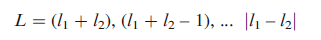
Energy states for which L=0, 1, 2, 3, 4... are known as S,P, D, F, G... terms, respectively. These are analogous to the s, p, d, f, g... labels used to denote atomic orbitals with l=0, 1, 2, 3, 4... in the 1-electron case.
NO molecule
The nitrogen atom has 2 + 5 = 7 electrons, and the oxygen atom has 2 + 6 = 8 electrons, making 15 electrons in the molecule.
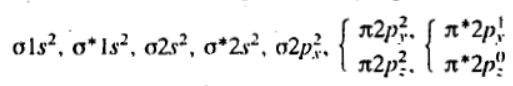
The inner shell is non-bonding. The bonding and antibonding 2s orbitals cancel, and a sigma bond is formed by the filled sigma 2px2 orbital. A pi bond is formed by the filled pi 2pz2 orbital. The half filled π*2py1 half cancels the filled π2py2 orbital, thus giving half a bond. The bond order is thus 2.5, that is in between a double and a triple bond. Alternatively the bond order may be worked out as (bonding - antibonding) /2, that is (10 - 5)/ = 2.5. The molecule is paramagnetic since it contains an unpaired electron.
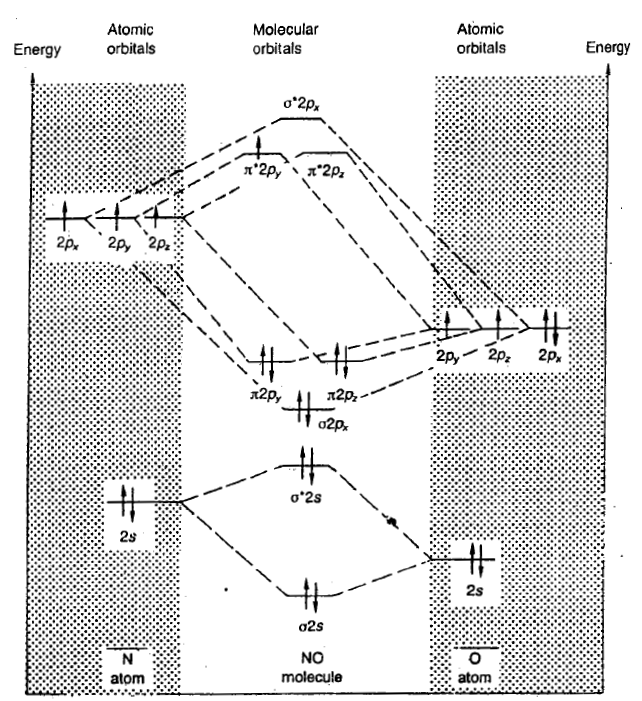
The only unpaired electron in the NO molecule is present in the π*2py orbital.
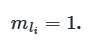
The total z-projection of L for the only unpaired electron in the π*2py orbital can be calculated as,