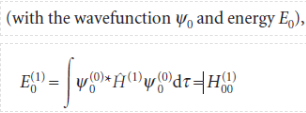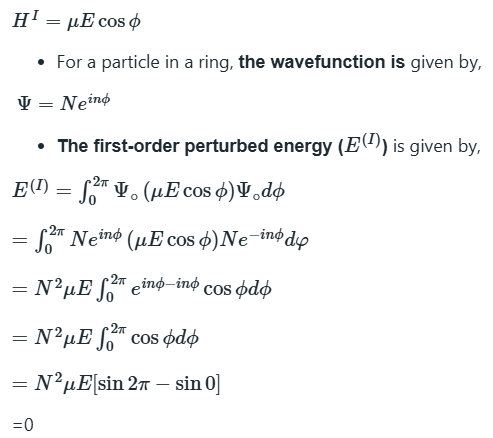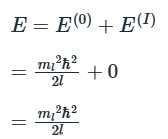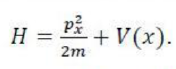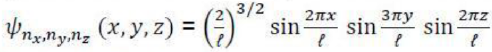Correct option is D
A particle on a ring
We consider a particle of mass m constrained to move in a circular path of radius r in the xy-plane. The total energy is equal to the kinetic energy, because V = 0 everywhere. We can therefore write E = p2/2m. According to classical mechanics, the angular momentum, Jz, around the z-axis (which lies perpendicular to the xy-plane) is Jz = ±pr, so the energy can be expressed as Jz2/2mr2. Because mr2 is the moment of inertia, I, of the mass on its path, it follows that
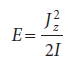
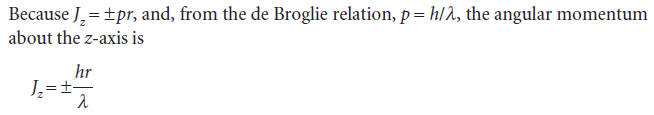
Opposite signs correspond to opposite directions of travel. This equation shows that the shorter the wavelength of the particle on a circular path of given radius, the greater the angular momentum of the particle.
Suppose for the moment that λ can take an arbitrary value. In that case, the wavefunction depends on the azimuthal angle φ. When φ increases beyond 2π, the wavefunction continues to change, but for an arbitrary wavelength it gives rise to a different value at each point, which is unacceptable. An acceptable solution is obtained only if the wavefunction reproduces itself on successive circuits. Because only some wavefunctions have this property, it follows that only some angular momenta are acceptable, and therefore that only certain rotational energies exist. Hence, the energy of the particle is quantized. Specifically, the only allowed wavelengths are
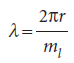
with ml, the conventional notation for this quantum number, taking integral values including 0. The value ml= 0 corresponds to λ =∞; a ‘wave’ of infinite wavelength has a constant height at all values of φ. The angular momentum is therefore limited to the values
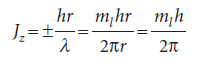
where we have allowed ml to have positive or negative values. That is,
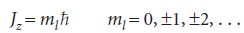
Positive values of ml correspond to rotation in a clockwise sense around the z-axis and negative values of ml correspond to counter-clockwise rotation around z.
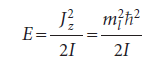
The corresponding normalized wavefunctions are
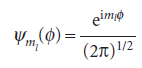
Time-independent perturbation theory

In time-independent perturbation theory, the perturbation is always present and unvarying. For example, it might represent a dip in the potential energy of a particle in a box in some region along the length of the box. In time-independent perturbation theory, we suppose that the true energy of the system differs from the energy of the simple system, and that we can write

The first-order corrections to the energy of the ground state