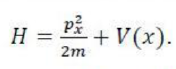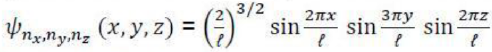Correct option is C
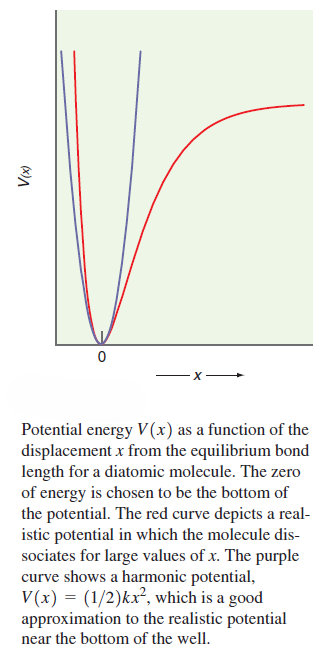
The energy needed to stretch the chemical bond can be described by a simple potential function such as that shown in Figure. The existence of a stable chemical bond implies that a minimum energy exists at the equilibrium bond length. The position of atoms in a molecule is dynamic rather than static. Think of the chemical bond as a spring rather than a rigid bar connecting the two atoms. Thermal energy increases the vibrational amplitude of the atoms about their equilibrium positions but does not change the vibrational frequency to a good approximation. The potential becomes steeply repulsive at short distances as the electron clouds of the atoms interpenetrate. It levels out at large distances because the overlap of electrons between the atoms required for chemical bond formation falls to zero.
The exact form of V(x) as a function of x depends on the molecule under consideration. The lowest one or two vibrational energy levels are occupied for most molecules for T~300K. Therefore, it is a good approximation to say that the functional form of the potential energy near the equilibrium bond length can be approximated by the harmonic potential.
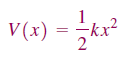
k is the force constant.
We expect the wave-particle of mass
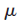
vibrating around its equilibrium distance to be described by a set of wave functions
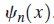
To find these wave functions and the corresponding allowed vibrational energies, the Schrödinger equation with the appropriate potential energy function must be solved:
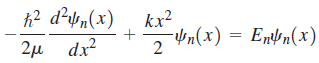
The solution of this second-order differential equation was well known in the mathematical literature from other contexts well before the development of quantum mechanics. We simply state that the normalized wave functions are
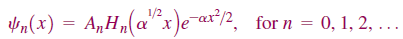
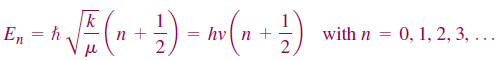
The energy stored in the quantum mechanical harmonic oscillator can only take on discrete values. The lowest state accessible to the system still has an energy greater than zero, referred to as a zero point energy. The frequency of oscillation is given by
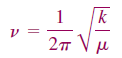
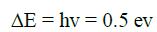
Zero point energy=
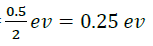

Minimum bond dissociation energy =4.75-0.25eV=4.50eV