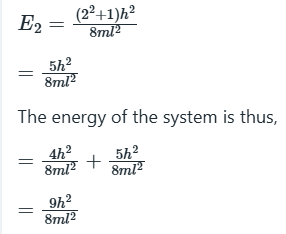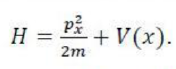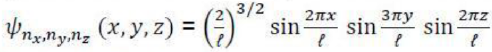Correct option is D
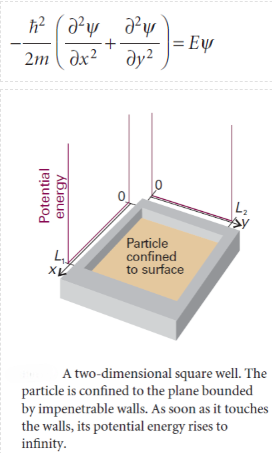
A particle is confined to a rectangular surface of length L1in the x-direction and L2in they-direction; the potential energy is zero everywhere except at the walls, where it is infinite. The wavefunction is now a function of both x and y and the Schrödinger equation is
Writing the wavefunction as a product of functions, one depending only on x and the other only on y:
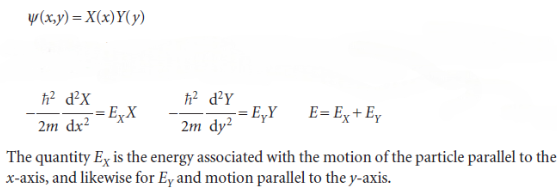
The first step in the justification of the separability of the wavefunction into the product of two functions X and Y is to note that, because X is independent of y and Y is independent of x, we can write
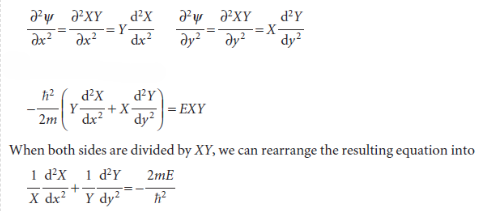
The first term on the left is independent of y, so if y is varied only the second term can change. But the sum of these two terms is a constant given by the right-hand side of the equation; therefore, even the second term cannot change when y is changed. In other words, the second term is a constant, which we write -2mEY/ℏ2. By a similar argument, the first term is a constant when x changes, and we write it -2mEX/ℏ2, and E = EX+ EY. Therefore, we can write
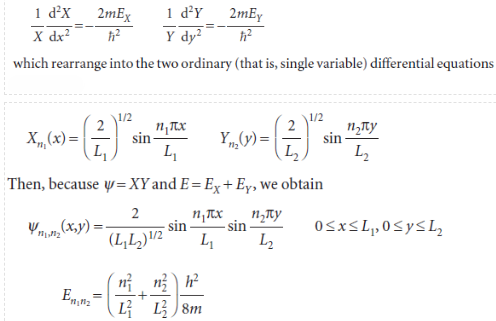
with the quantum numbers taking the values n1 =1, 2, . . . and n2 =1, 2, . . . independently.
Degeneracy
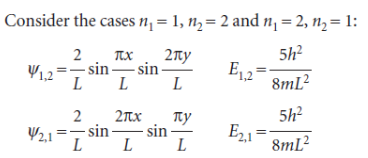
We see that, although the wavefunctions are different, they are degenerate, meaning that they correspond to the same energy. In this case, in which there are two degenerate wavefunctions, we say that the energy level 5(h2/8mL2) is ‘doubly degenerate’.
In the ground state, the two electrons can be placed in an energy state: (1,1)
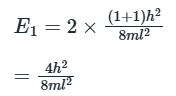
In the first excited state, one electron can be placed in a doubly degenerate energy level.