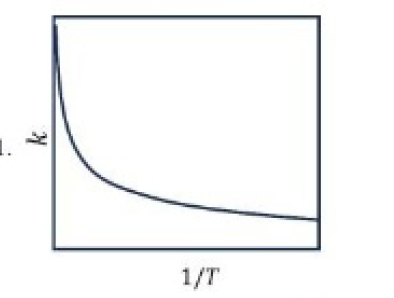
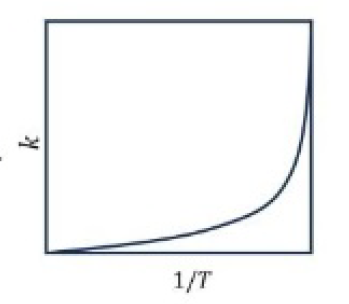
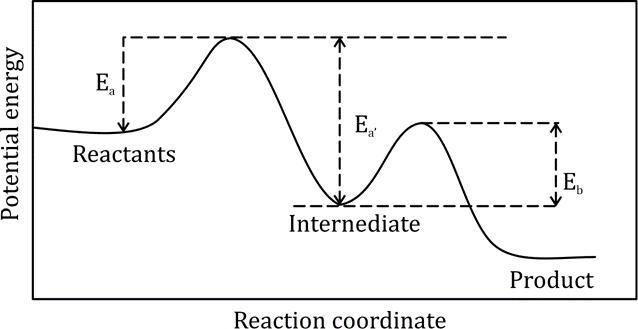
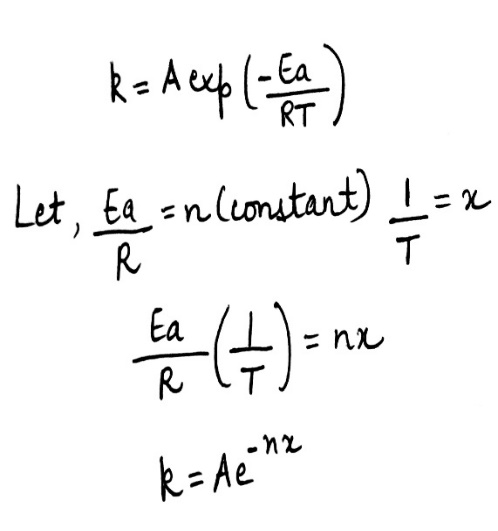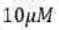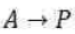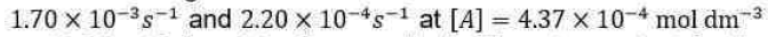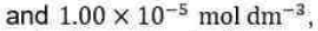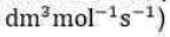Correct option is A


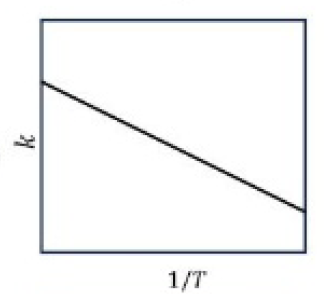
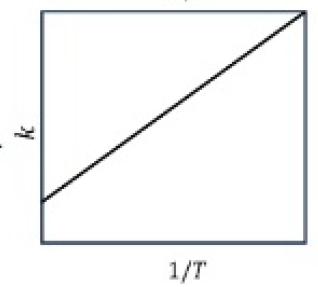
The main characteristics of Arrhenius equation are described below.
1.Larger the activation energy, smaller the value of rate constant.
2.Larger the activation energy, greater the effect of a given temperature rise on k.
3.At lower temperatures, increase in temperature causes more change in the value of k than that at higher temperatures.