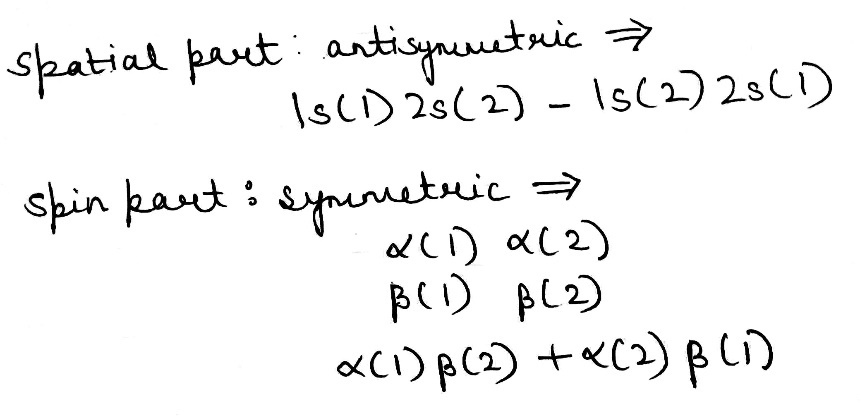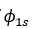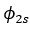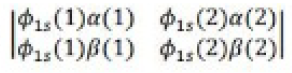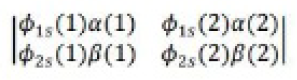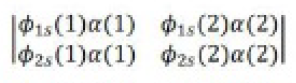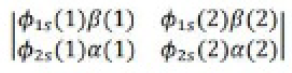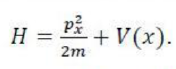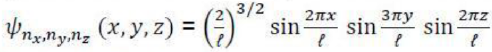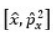Correct option is C
It was proposed by J.C. Slater in 1930 that for a multielectron atom the total wave function must consist of the spatial part and the spin part. He showed that the total atomic wave function, which is the product of the spatial wave function and the spin wave function, is antisymmetric (A) with respect to the interchange of electrons, i.e.


This result follows from the fact that if in a determinant two rows (or columns) are interchanged, it changes sign. What is true of a two-electron system is also true of polyelectron atoms and molecules. The polyelectron wave function also satisfies the antisymmetry rule. The solution of the Schrödinger wave equation for such systems must start from the appropriate Slater determinant for a given electronic configuration.