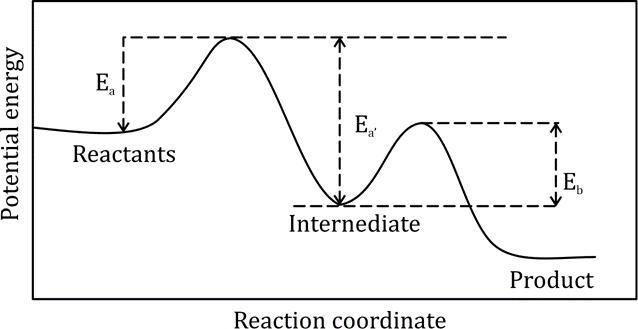
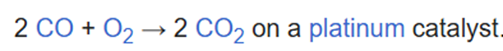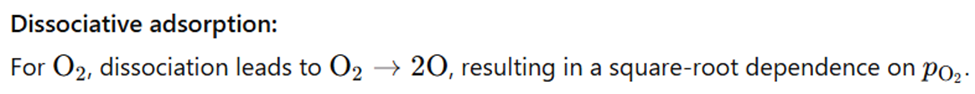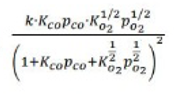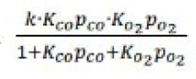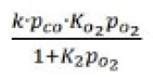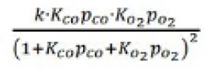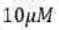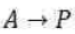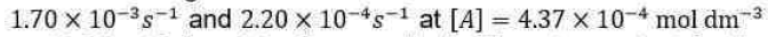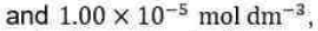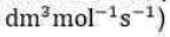Correct option is A
Reactions on surfaces are reactions in which at least one of the steps of the reaction mechanism is the adsorption of one or more reactants. The mechanisms for these reactions, and the rate equations are of extreme importance for heterogeneous catalysis.


Langmuir–Hinshelwood mechanism
In this mechanism, two molecules adsorb on neighboring sites and the adsorbed molecules undergo a bimolecular reaction: