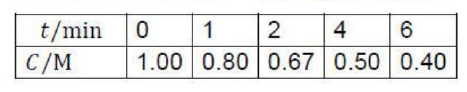
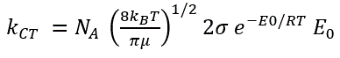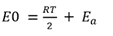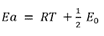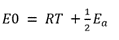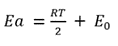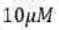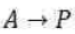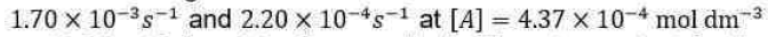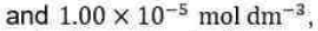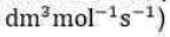Correct option is D
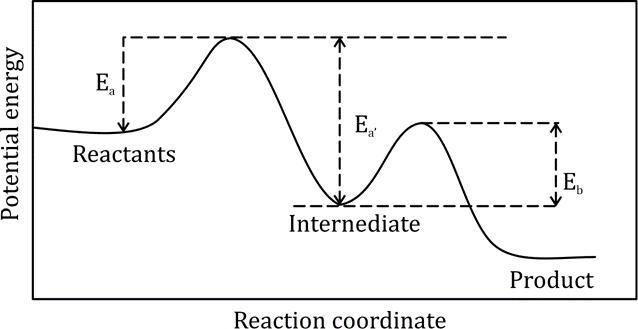
Collision theory is a principle of chemistry used to predict the rates of chemical reactions. It states that when suitable particles of the reactant hit each other with the correct orientation, only a certain amount of collisions result in a perceptible or notable change; these successful changes are called successful collisions. The successful collisions must have enough energy, also known as activation energy, at the moment of impact to break the pre-existing bonds and form all new bonds. This results in the products of the reaction.
The rate for a bimolecular gas-phase reaction, A + B → product, predicted by collision theory is
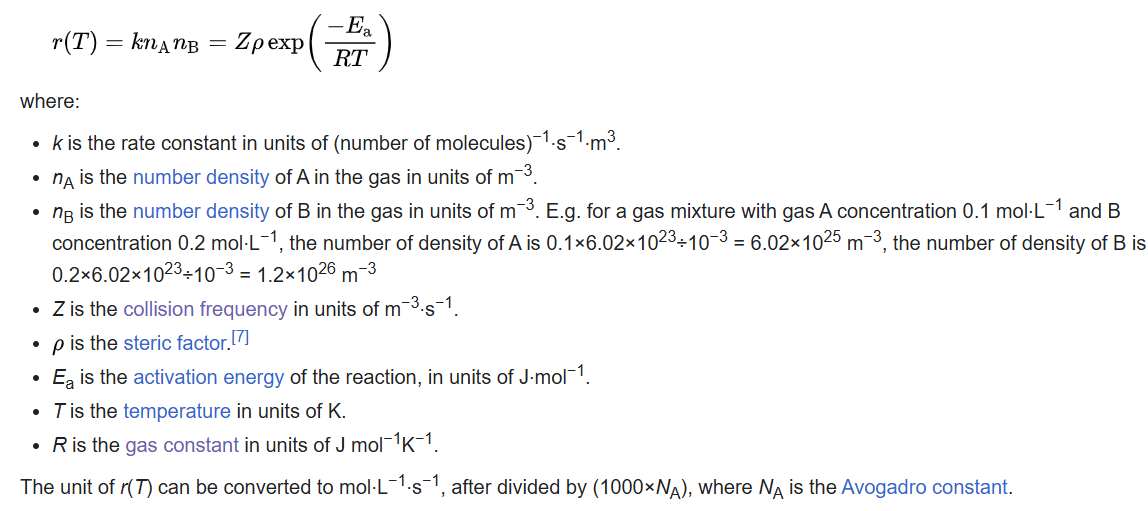
For a reaction between A and B, the collision frequency calculated with the hard-sphere model with the unit number of collisions per m3 per second is:
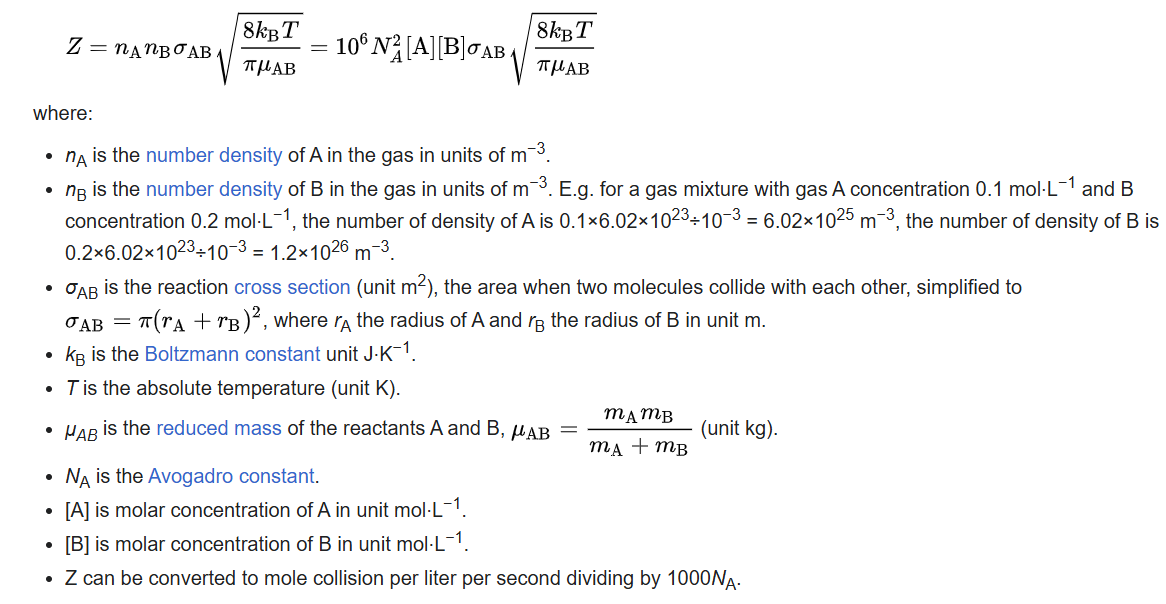
If all the units that are related to dimension are converted to dm, i.e. mol⋅dm-3 for [A] and [B], dm2 for σAB , dm2 ⋅kg⋅s-2 ⋅K-1 for the Boltzmann constant, then
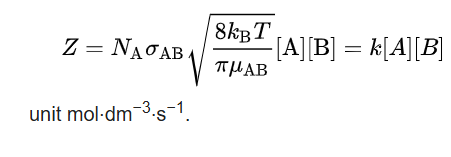
In physical chemistry, the Arrhenius equation is a formula for the temperature dependence of reaction rates.


The only difference is the unit of Ea: the former form uses energy per mole, which is common in chemistry, while the latter form uses energy per molecule directly, which is common in physics. The different units are accounted for in using either the gas constant, R, or the Boltzmann constant, kB , as the multiplier of temperature T.