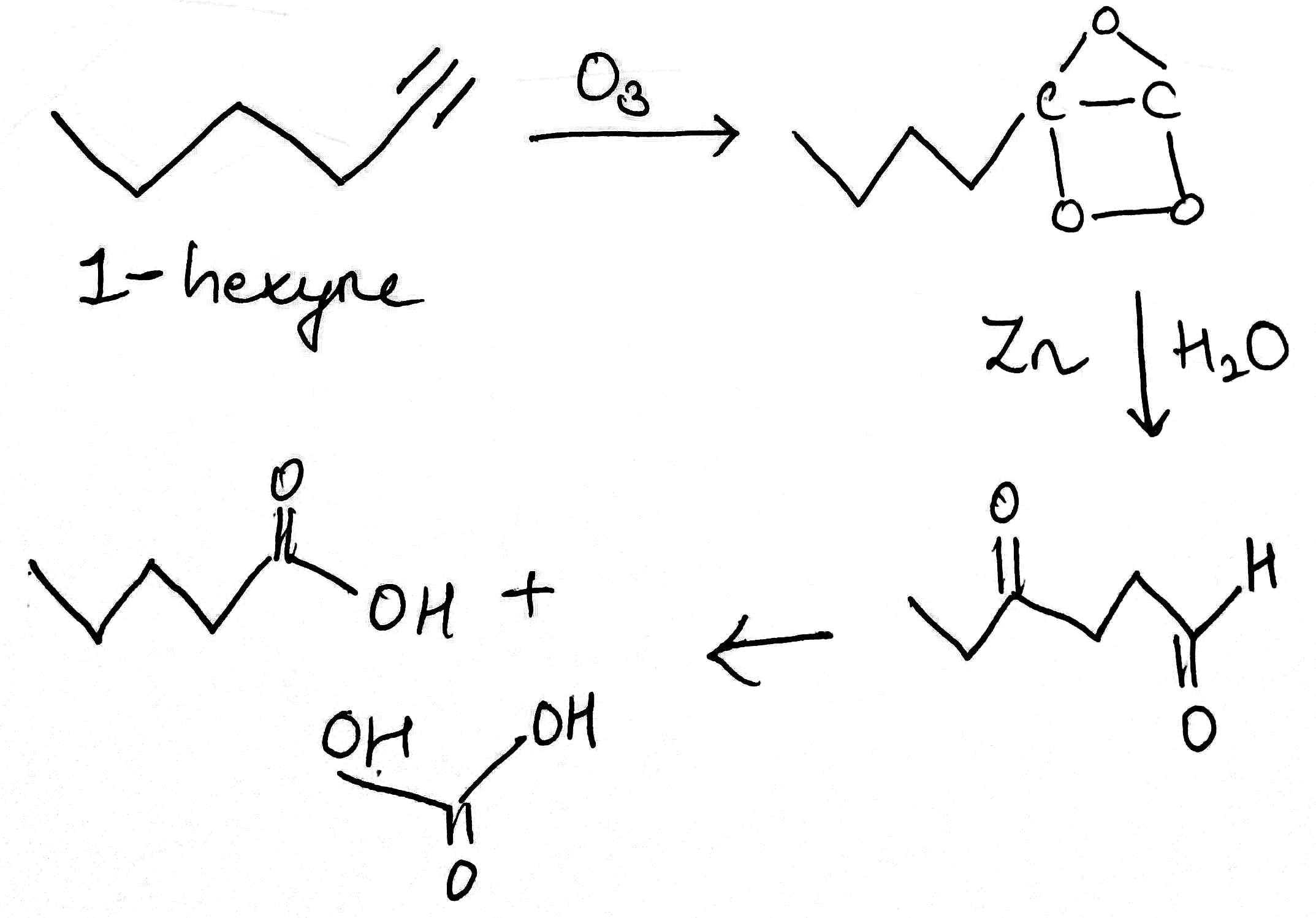
Correct option is B
Consider the compound to be X.
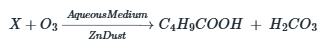
The parent hydrocarbon must have six carbons.
Information Booster
Ozonolysis is an organic reaction where the unsaturated bonds are cleaved with ozone (O3). Multiple carbon–carbon bond are replaced by carbonyl (C=O) groups, such as aldehydes, ketones, and carboxylic acids.
The use of triphenylphosphine, thiourea, zinc dust, or dimethyl sulfide produces aldehydes or ketones. While the use of sodium borohydride produces alcohols. (R group can also be hydrogens). The use of hydrogen peroxide can produce carboxylic acids.
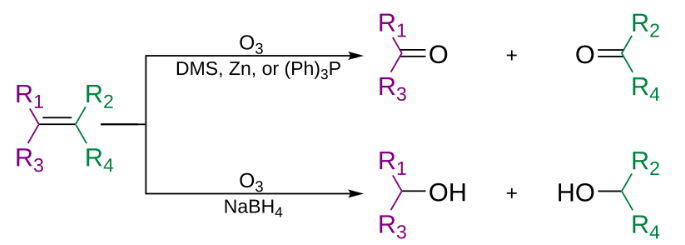
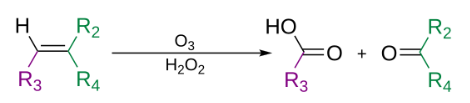
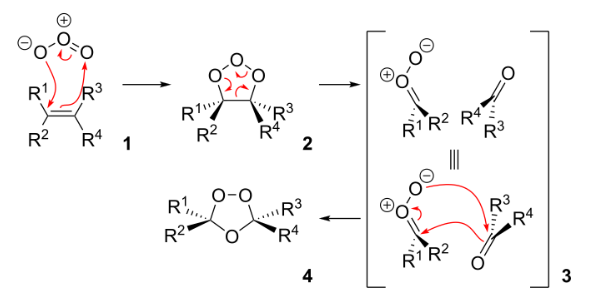
In the generally accepted mechanism, the alkene and ozone form an intermediate molozonide in a 1,3-dipolar cycloaddition. Next, the molozonide reverts to its corresponding carbonyl oxide (also called the Criegee intermediate or Criegee zwitterion) and aldehyde or ketone (3) in a retro-1,3-dipolar cycloaddition. The oxide and aldehyde or ketone react again in a 1,3-dipolar cycloaddition, producing a relatively stable ozonide intermediate, known as a trioxolane (4).