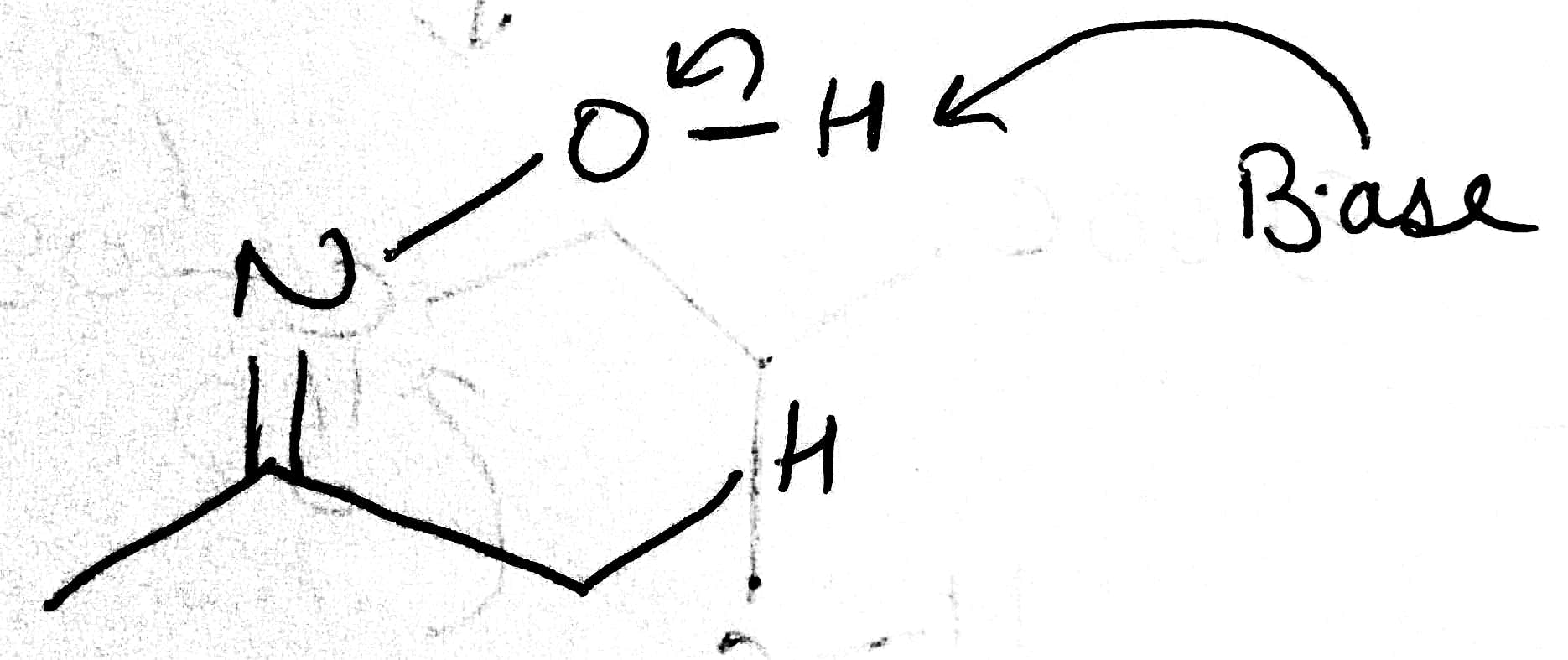
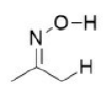
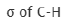Correct option is A
A base will abstract the most acidic hydrogen. A negative charge will be more stable on the oxygen atom (due to its high electronegativity) than on the carbon atom.
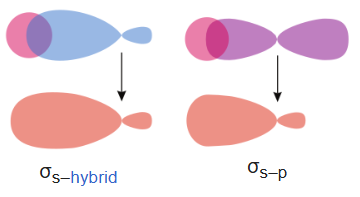
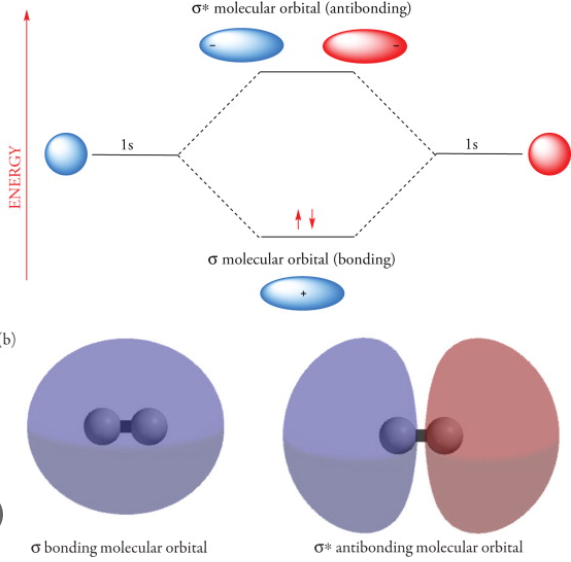
Sigma bonds (σ bonds) or sigma overlap are the strongest type of covalent chemical bond. They are formed by head-on overlapping between atomic orbitals along the internuclear axis.
For homodiatomics (homonuclear diatomic molecules), bonding σ orbitals have no nodal planes at which the wavefunction is zero, either between the bonded atoms or passing through the bonded atoms. The corresponding antibonding, or σ* orbital, is defined by the presence of one nodal plane between the two bonded atoms.