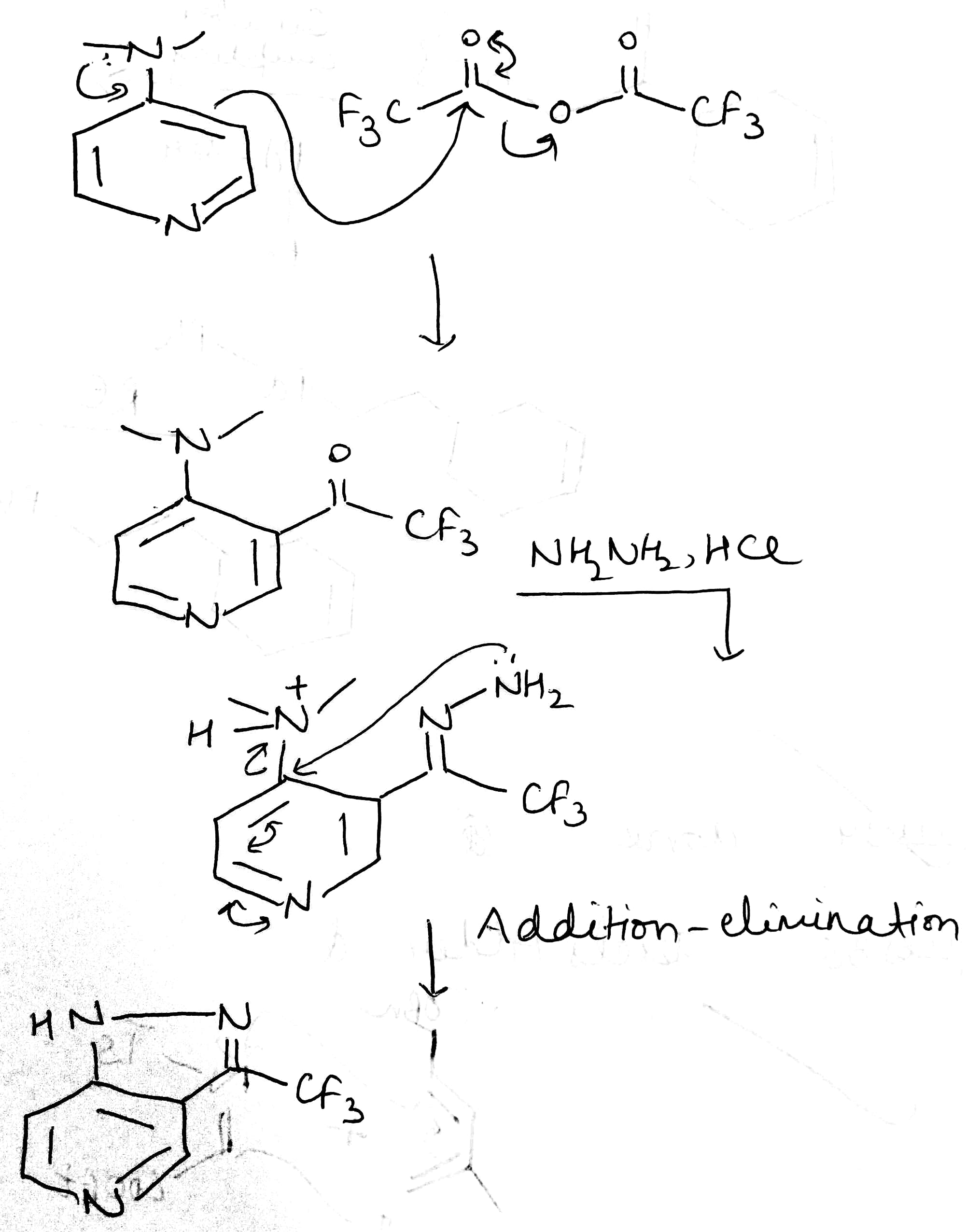
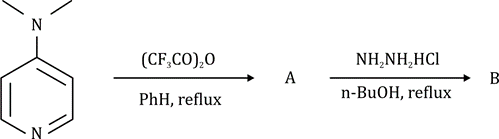
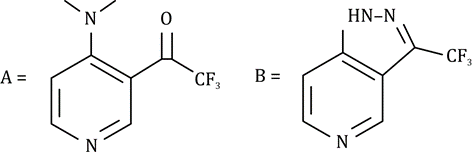
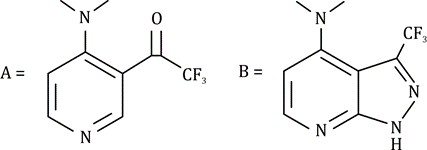
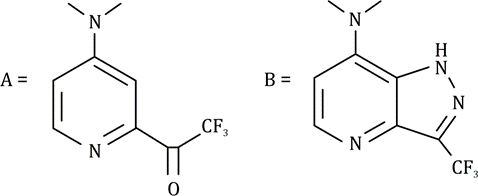
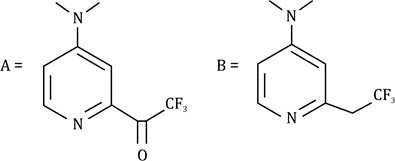
Correct option is A
The addition-elimination mechanism
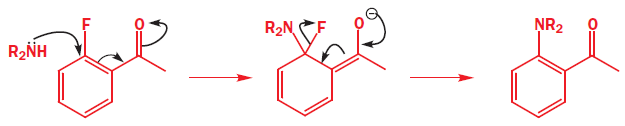
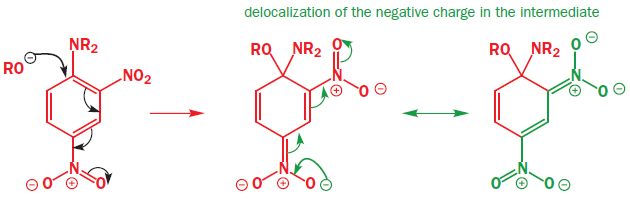
The mechanism involves addition of the nucleophile followed by elimination of the leaving group—the addition–elimination mechanism. It is not necessary to have a carbonyl group—any electron-withdrawing group will do—the only requirement is that the electrons must be able to get out of the ring into this anion-stabilizing group. Here is an example with a para-nitro group.
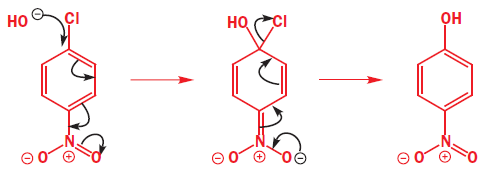
Everything is different about this example—the nucleophile (HO-), the leaving group (Cl-), the anion-stabilizing group (NO2), and its position (para)—but the reaction still works. The nucleophile is a good one, the negative charge can be pushed through on to the oxygen atom(s) of the nitro group, and chloride is a better leaving group than OH.
A typical nucleophilic aromatic substitution has:
a. an oxygen, nitrogen, or cyanide nucleophile
b. a halide for a leaving group
c. a carbonyl, nitro, or cyanide group ortho or para to the leaving group
Here is an example with RO- attacking a nitrated aniline.