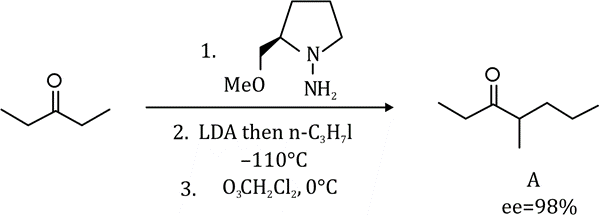
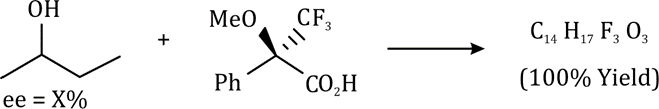
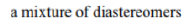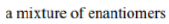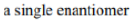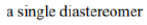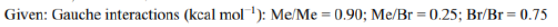Correct option is A
The most important protecting groups for alcohols are ethers and mixed acetals.
Acetals

The THP group is a widely used protecting group; it is readily introduced by reaction of the enol ether dihydropyran with an alcohol in the presence of an acid catalyst, such as TsOH,
For sensitive alcohols such as allylic alcohols, PPTS (pyridinium p-toluenesulfonate) is used as a catalyst for tetrahydropyranylation. As an acetal, the THP group is readily hydrolyzed under aqueous acidic conditions with AcOH-THF, TsOH, PPTS-EtOH, or Dowex-H (cation exchange resin).
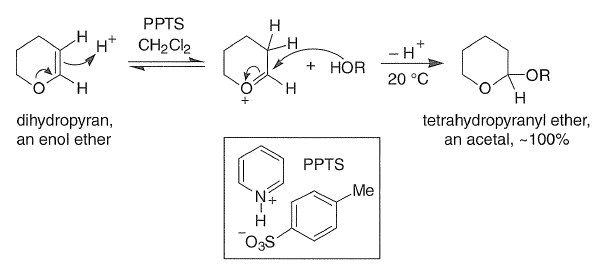
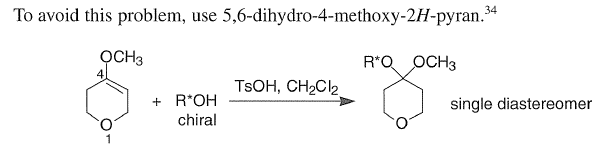
Protection of chiral alcohols as THP ethers leads to mixtures of diastereoisomers.
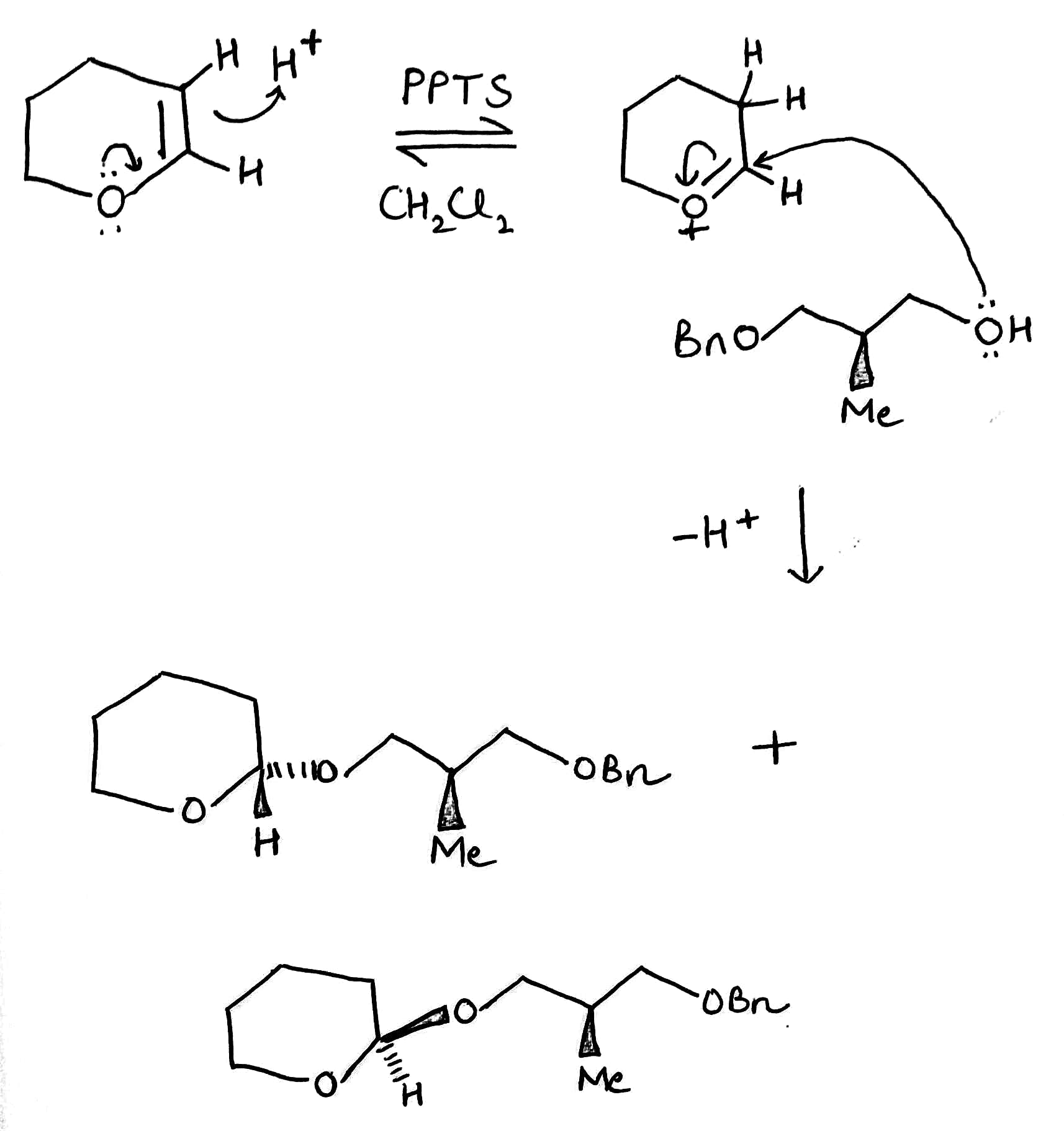
Diastereomers (sometimes called diastereoisomers) are a type of stereoisomer. Diastereomers are defined as non-mirror image, non-identical stereoisomers. Hence, they occur when two or more stereoisomers of a compound have different configurations at one or more (but not all) of the equivalent (related) stereocenters and are not mirror images of each other.
Diastereomers differ from enantiomers in that the latter are pairs of stereoisomers that differ in all stereocenters and are therefore mirror images of one another.