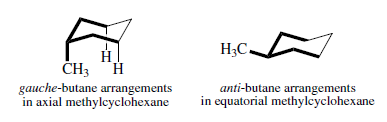Correct option is B
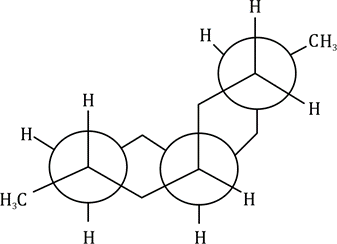
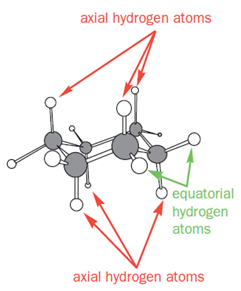
In the chair conformation of cyclohexane, all six carbon atoms are identical, but there are two types of protons-one type stick either vertically up or down and are called axial hydrogen atoms; the other sort stick out sideways and are called equatorial hydrogen atoms.
As you go round the ring, notice that each of the CH2 groups has one hydrogen sticking up and one sticking down. However, all the ‘up’ ones alternate between axial and equatorial, as do all the ‘down’ ones.
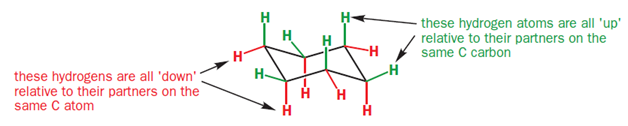
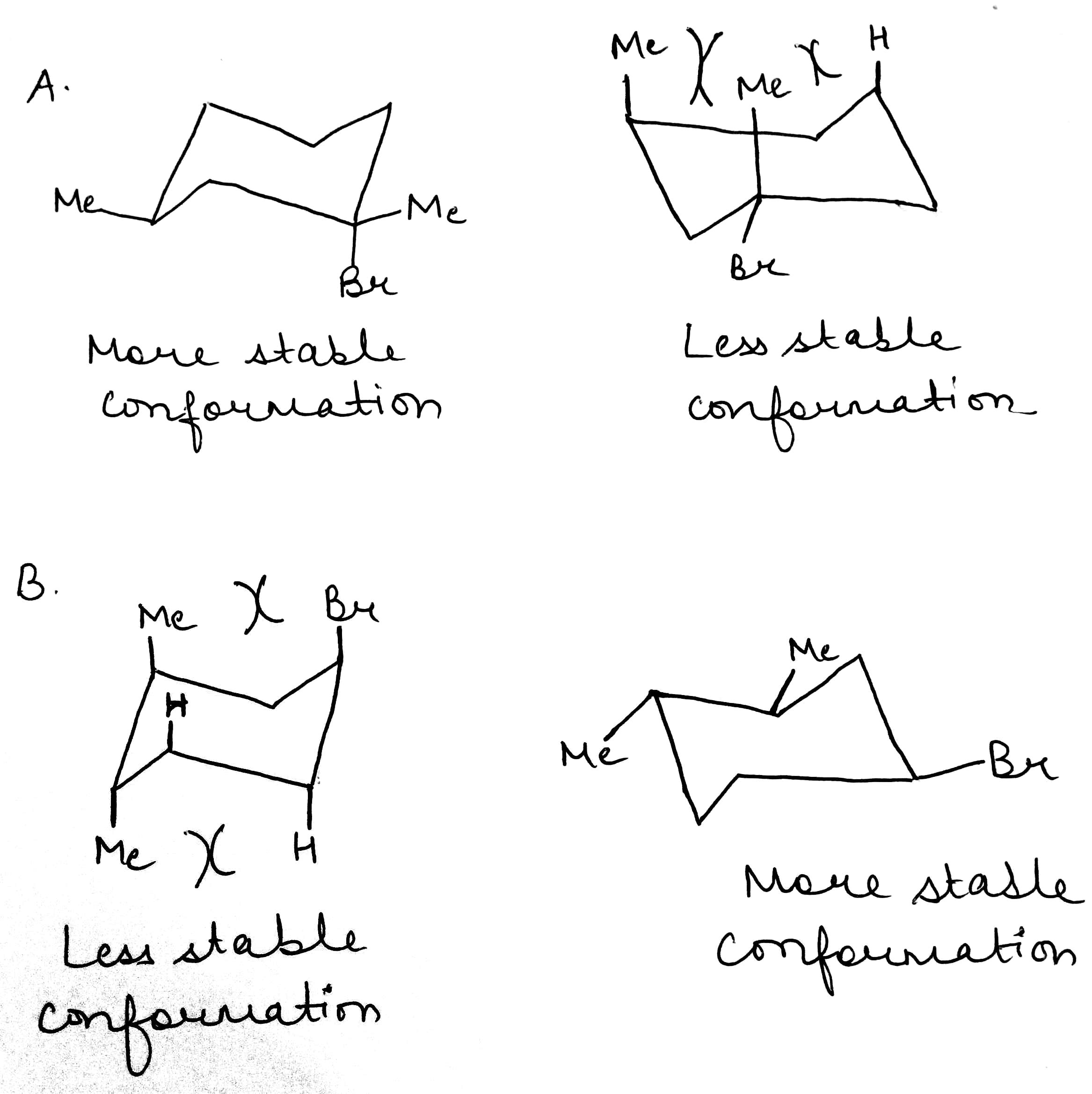
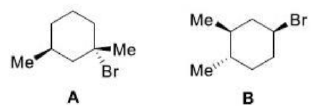
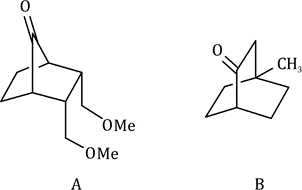
In a monosubstituted cyclohexane, there can exist two different chair conformers: one with the substituent axial, the other with it equatorial. When there are two or more substituents on the ring, stereoisomerism is possible.
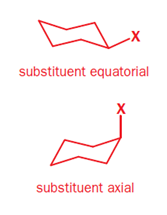
Remember, the ‘cis’ and ‘trans’ prefixes merely indicate that both groups are on the same ‘side’ of the cyclohexane ring. Whether the substituents are both axial/equatorial or one axial and the other equatorial depends on the substitution pattern. Each time you meet a molecule, draw the conformation or make a model to find out which bonds are axial and equatorial.
It is not always easy to decide if an equatorial substituent is ‘up’ or ‘down’. The key is to compare it with its axial partner on the same C atom-axial substituents very clearly point ‘up’ or ‘down’. If the axial partner is ‘up’, the equatorial substituent must be ‘down’ and vice versa.
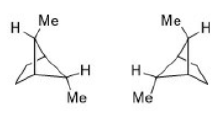
Substitution on a cyclohexane ring does not greatly affect the rate of conformational inversion but does change the equilibrium distribution between alternative chair forms. All substituents that are axial in one chair conformation become equatorial on ring inversion, and vice versa. For methylcyclohexane, ΔG° for the equilibrium
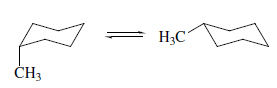
is - 1.8 kcal/mol, corresponding to a composition with 95% of the equatorial methyl conformation. Two factors contribute to the preference for the equatorial conformation. The equatorial methyl conformation corresponds to an anti arrangement with respect to the C(2)-C(3) and C(6)-C(5) bonds, whereas the axial methyl group is in a gauche relationship to these bonds. Gauche conformation of n-butane is 0.8 kcal/mol higher in energy than the anti conformation. In addition, there is a Vander Waals repulsion between the axial methyl group and the axial hydrogens at C-3 and C-5. Interactions of this type are called 1,3-diaxial interactions.