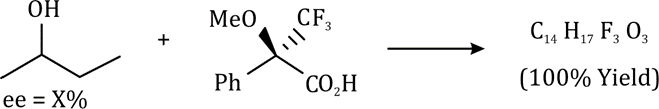
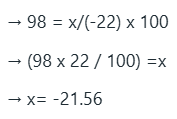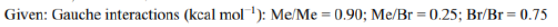Correct option is C
In chemistry, specific rotation ([α]) is a property of a chiral chemical compound. It is defined as the change in orientation of monochromatic plane-polarized light, per unit distance–concentration product, as the light passes through a sample of a compound in solution. Compounds which rotate the plane of polarization of a beam of plane polarized light clockwise are said to be dextrorotary, and correspond with positive specific rotation values, while compounds which rotate the plane of polarization of plane polarized light counterclockwise are said to be levorotary, and correspond with negative values. If a compound is able to rotate the plane of polarization of plane-polarized light, it is said to be “optically active”.
Specific rotation is an intensive property, distinguishing it from the more general phenomenon of optical rotation. As such, the observed rotation (α) of a sample of a compound can be used to quantify the enantiomeric excess of that compound, provided that the specific rotation ([α]) for the enantiopure compound is known.
If the specific rotation,
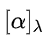
of a pure chiral compound is known, it is possible to use the observed specific rotation,
to determine the enantiomeric excess (ee), or "optical purity", of a sample of the compound, by using the formula:
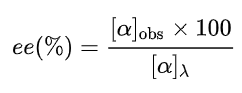
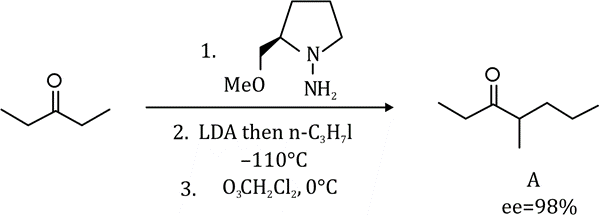
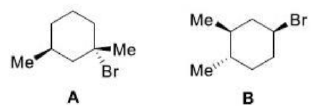
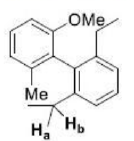
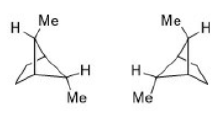
The given product is chiral. Chiral molecules will usually have a stereogenic element from which chirality arises. The most common type of stereogenic element is a stereogenic center, or stereocenter. In the case of organic compounds, stereocenters most frequently take the form of a carbon atom with four distinct (different) groups attached to it in a tetrahedral geometry.