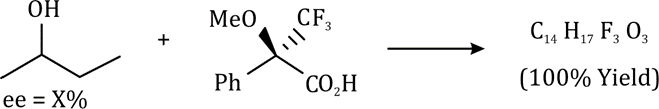
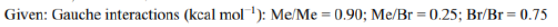Correct option is C
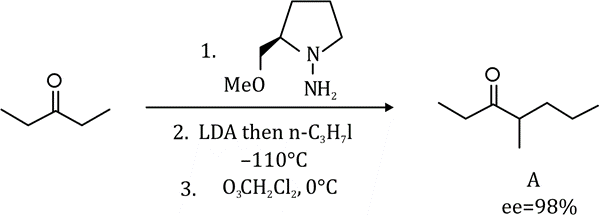
In stereochemistry, enantiomeric excess (ee) is a measurement of purity used for chiral substances. It reflects the degree to which a sample contains one enantiomer in greater amounts than the other. A racemic mixture has an ee of 0%, while a single completely pure enantiomer has an ee of 100%. A sample with 70% of one enantiomer and 30% of the other has an ee of 40% (70% − 30%).
If one knows the moles of each enantiomer produced then:
% ee = ((R–S)/(R+S)×100)
3R : 2S
R = 2S/3
given: R% + S% =100%
calculation:
Putting the value of R in above eq.
2S/3 + S = 100
(2S + 3S)/ 3 =100
5S/3 = 100
S = 60%
R = 40%
Therefore, ee% = 60% –40%
ee% = 20%