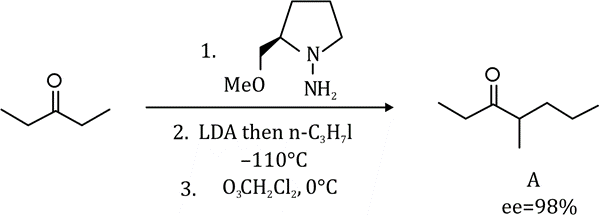
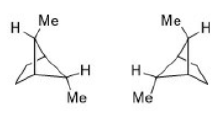
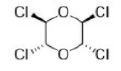
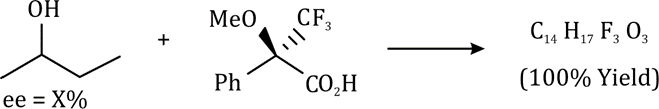
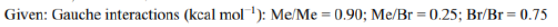Correct option is C
Certain compounds that do not contain asymmetric atoms are nevertheless chiral. For these compounds, we can draw two perpendicular planes neither of which can be bisected by a plane of symmetry. If either plane could be so bisected, the molecule would be superimposable on its mirror image, since such a plane would be a plane of symmetry.
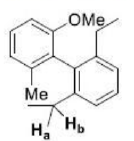
Biphenyls containing four large groups in the ortho positions cannot freely rotate about the central bond because of steric hindrance.
Enantiotopic and Diastereotopic atoms
Many molecules contain atoms or groups that appear to be equivalent, but with a close inspection will show to be actually different. We can test whether two atoms are equivalent by replacing each of them in turn with some other atom or group. If the new molecules created by this process are identical, the original atoms are equivalent; otherwise they are not.
In the case of ethanol CH2MeOH, if we replace one of the CH2 hydrogens by a group Z, we get one enantiomer of the compound ZCHMeOH, while replacement of the other hydrogen gives the other enantiomer. Since the two compounds that result upon replacement of H by Z are not identical but enantiomeric, the hydrogens are not equivalent. We define as enantiotopic two atoms or groups that upon replacement with a third group give enantiomers.
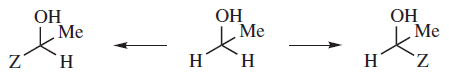
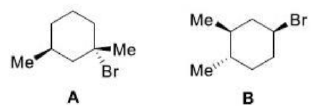
Where two atoms or groups in a molecule are in such positions that replacing each of them in turn by a group Z gives rise to diastereomers, the atoms or groups are called diastereotopic.
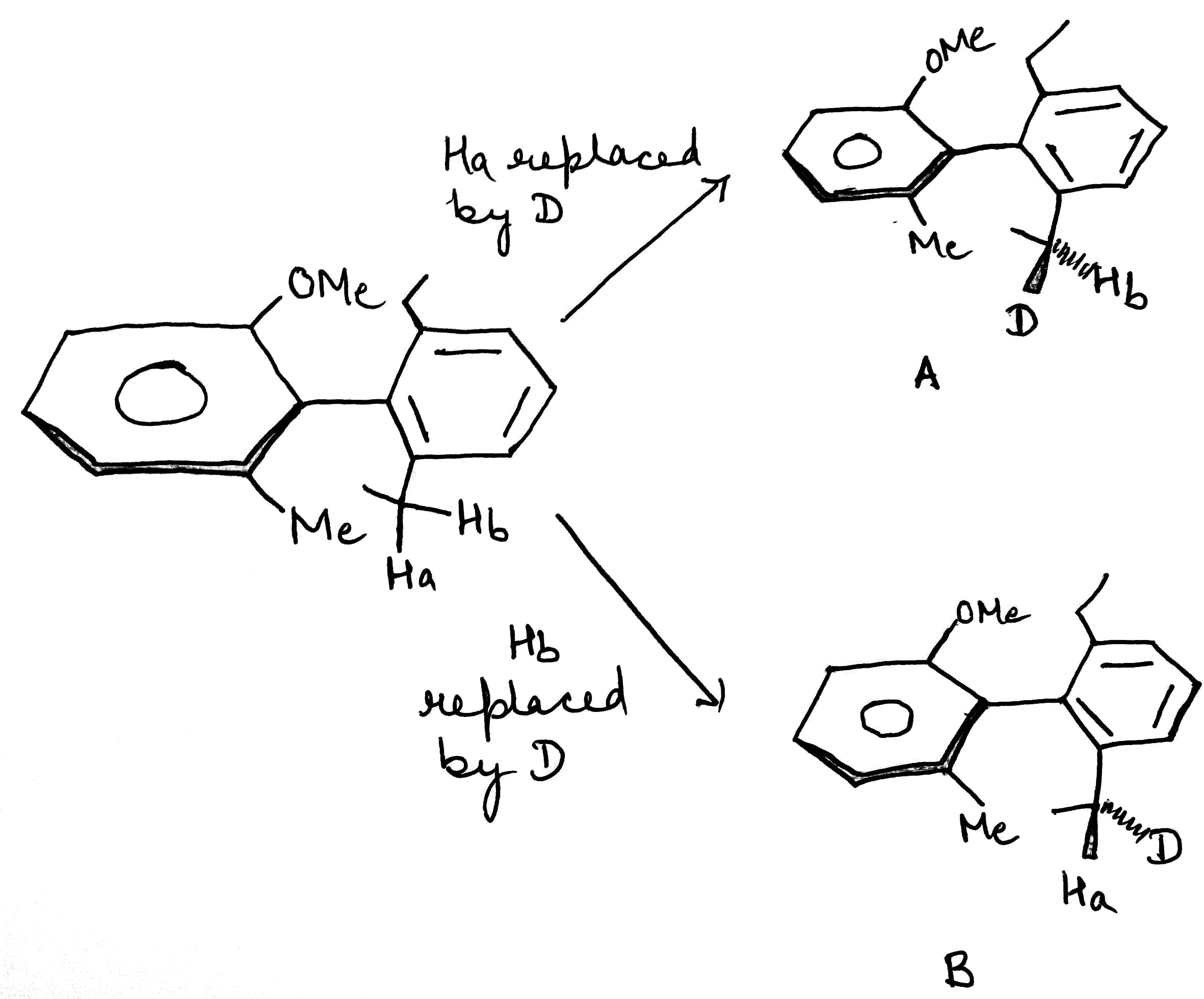
A and B do not have mirror image relationship.
Information Booster
Identical compounds (homomers) are those in which all of the atoms are arranged in the same spatial orientation.
Enantiomers
An enantiomer is one of a pair of molecular entities which are mirror images of each other and non-superposable. Enantiomer molecules are like right and left hands: one cannot be superposed onto the other without first being converted to its mirror image. It is solely a relationship of chirality and the permanent three-dimensional relationships among molecules or other chemical structures: no amount of re-orientation of a molecule as a whole or conformational change converts one chemical into its enantiomer.
A common naming conventions for specifying one of the two enantiomers (the absolute configuration) of a given chiral molecule: the R/S system is based on the geometry of the molecule. A pair of enantiomers have opposite configurations.
Diastereomers
In stereochemistry, diastereomers are a type of stereoisomer. Diastereomers are defined as non-mirror image, non-identical stereoisomers. Hence, they occur when two or more stereoisomers of a compound have different configurations at one or more (but not all) of the equivalent (related) stereocenters and are not mirror images of each other. When two diastereoisomers differ from each other at only one stereocenter, they are epimers. Each stereocenter gives rise to two different configurations and thus typically increases the number of stereoisomers by a factor of two.
Diastereomers differ from enantiomers in that the latter are pairs of stereoisomers that differ in all stereocenters and are therefore mirror images of one another. Enantiomers of a compound with more than one stereocenter are also diastereomers of the other stereoisomers of that compound that are not their mirror image (that is, excluding the opposing enantiomer).
Constitutional isomers
In chemistry, a structural isomer (or constitutional isomer in the IUPAC nomenclature) of a compound is another compound whose molecule has the same number of atoms of each element, but with logically distinct bonds between them.