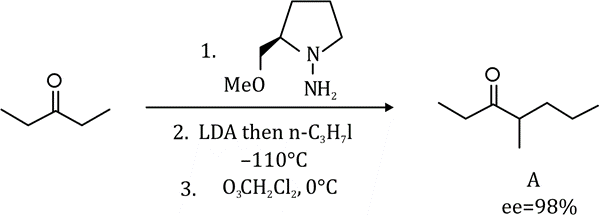
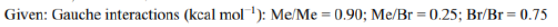Correct option is A
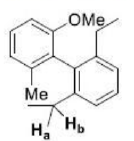
Enantiotopic faces in a chemical compound are non-equivalent faces, i.e., two faces A and B are enantiotopic if the molecule gives rise to enantiomeric pair when the faces are attacked with some reagent (such as Cl-) while the remaining parts of the molecule stay fixed. Enantiotopic faces are always identical in the achiral environment and different in chiral media. For instance, two faces of primary carbocation are homotopic, as the attack on two faces by an incoming nucleophile generates enantiomeric pair.
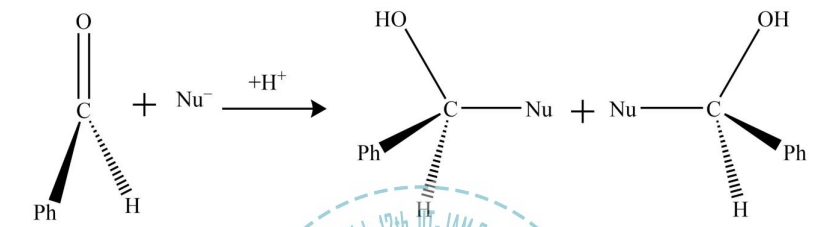
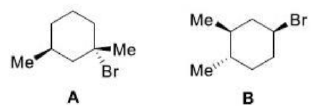
Diastereotopic faces in a chemical compound are non-equivalent faces, i.e., two faces A and B are diastereotopic if the molecule gives rise to diastereotopic pair when the faces are attacked with some reagent (such as Cl-) while the remaining parts of the molecule stay fixed. Diastereotopic faces are always non-identical in any type of environment whether it is chiral or achiral. For instance, two faces of 2-methylbutanal are diastereotopic, as the attack on two faces by an incoming nucleophile generates diastereomeric pair.
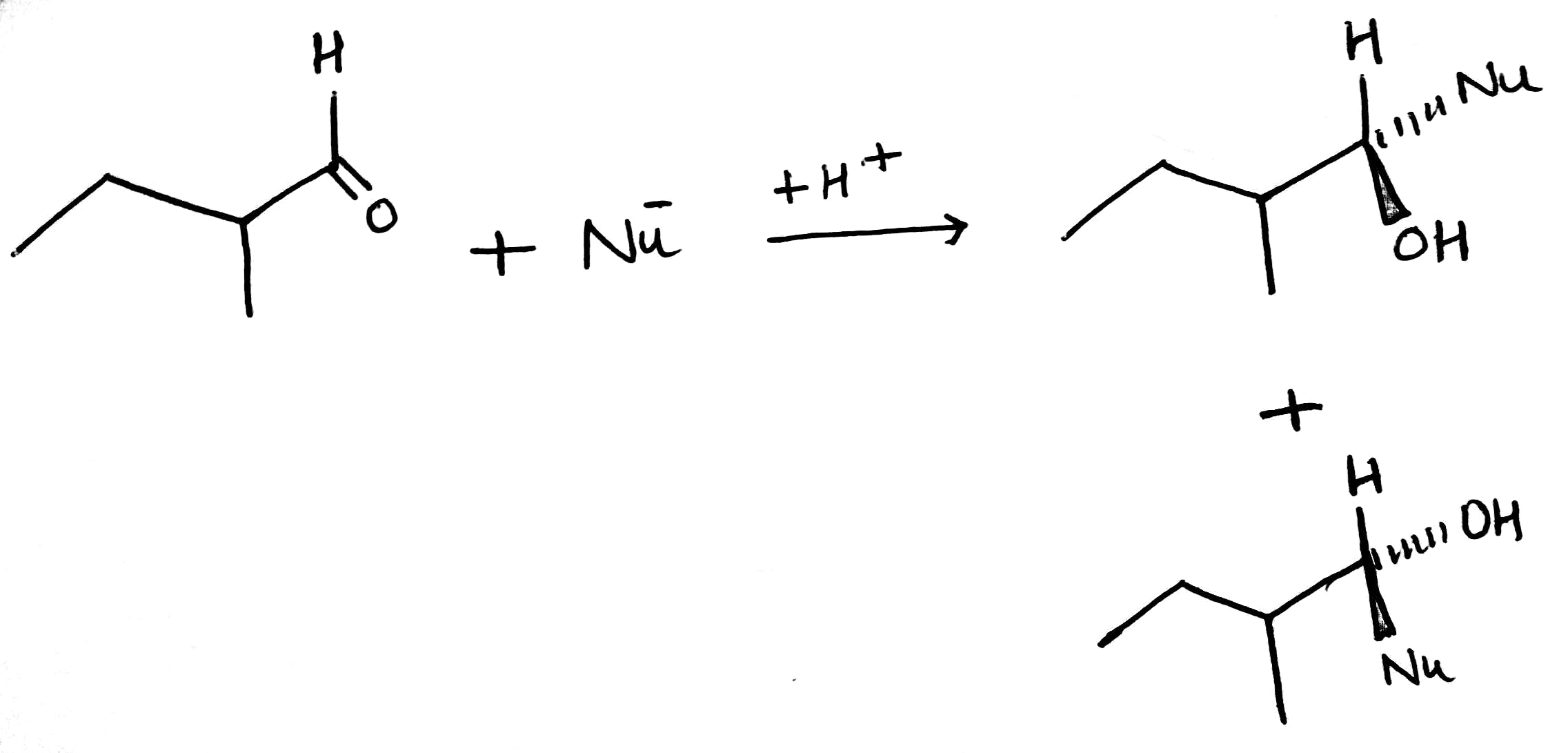
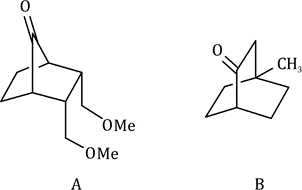
Structure A has sp3 hybridised carbon atoms.
Structure B: A plane can be passed through the centre of the bridge.