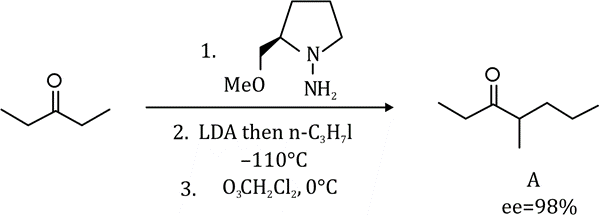
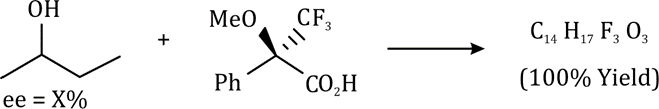
Correct option is C
Nuclear magnetic resonance (NMR) spectroscopy is a resonance technique involving absorption of radiofrequency energy. The magnetic environment of a nucleus affects its resonance frequency and allows structural information to be deduced. NMR-active nuclei, particularly those with a spin quantum number of 1/2, are of great significance in NMR spectroscopy. Examples include1H,13C,15N, and31P.
Shielded and Deshielded Protons
The valence-shell electron densities vary from one proton to another. In an applied magnetic field, i.e., when the molecule is placed in a uniform external magnetic field, the valence electrons around the protons are induced to circulate and this circulation, in turn, generates a secondary magnetic field, i.e., induced magnetic field. Circulation of electrons (specially πelectrons) about the nearby nuclei generates a field that can either oppose or reinforce the applied field at that proton. If the induced field opposes the applied field, the proton feels a lower field strength, and then the proton is said to be shielded. But if the induced field reinforces the applied field, the proton feels a higher field strength, and thus, such a proton is said to be deshielded.
Therefore, the different protons in a molecule do not have resonance at exactly the same frequency. This variability is due to the fact that the protons in a molecule are surrounded by electrons and exist in slightly different electronic (magnetic) environments from one another. The protons are shielded or deshielded by the electrons that surround them.
To generate an NMR spectrum, magnetic field strength (B0) is increased from left to right. The signal for the reference compound tetramethyl silane (TMS) appears at the extreme right of the spectrum with δ = 0 ppm. Due to the low electronegativity of silicon, the shielding of equivalent protons in TMS is greater than most of the organic compounds. Consequently, for most of the organic molecules, signals of the protons appear to the left of TMS signal in the NMR spectra. The more deshielded proton signals appear at the higher δ value, lower field (downfield) which is towards the left side of the plot. The more shielded proton signals appear at the lower δ value, higher field (upfield) which is towards the right side of the plot.
Spin-spin coupling
Structural assignment is often helped by the observation of the spin–spin coupling, which gives rise to multiplets in the spectrum due to interactions between nuclear spins. Spin–spin coupling arises when the orientation of the spin of a nearby nucleus affects the energy of another nucleus and causes small changes in the location of the latter’s resonance. A multiplet of 2I+1 lines is obtained when a spin-1/2 nucleus (or a set of symmetry-related spin-1/2 nuclei) is coupled to a nucleus of spin I. The coupling of the nuclear spins of different elements is called heteronuclear coupling. Homonuclear coupling between nuclei of the same element is detectable when the nuclei are in chemically inequivalent locations.
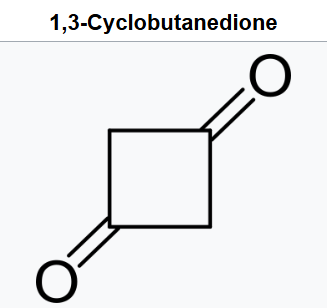
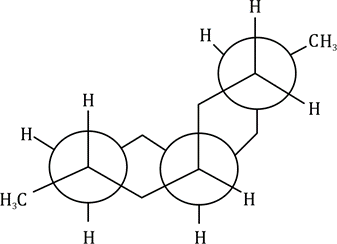
In the above image, two different type of carbon atoms are present resulting in two different signals.
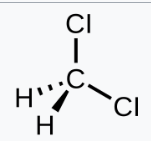
Dichloromethane will give one signal. But due to the presence of electronegative atoms, the signal in proton NMR will be deshielded (more than the given value)
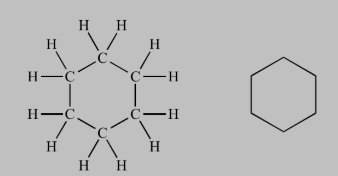
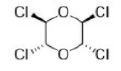
All the carbon atoms are identical and will give one signal. A single peak will also be observed for the all the identical protons.