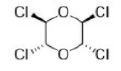
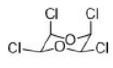
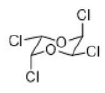
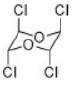
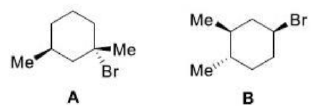
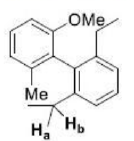
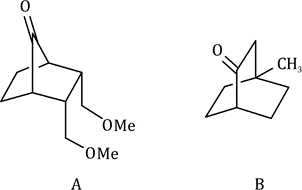
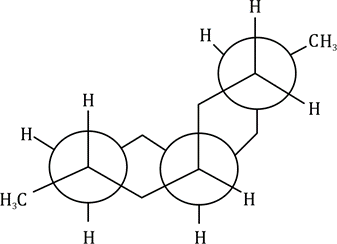
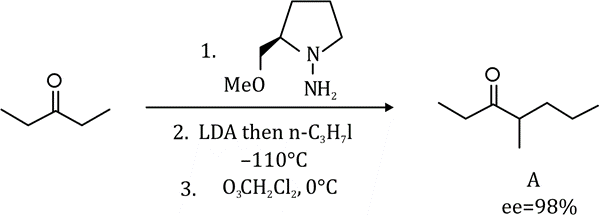
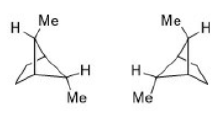
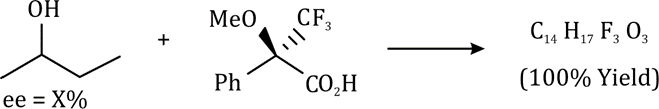
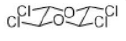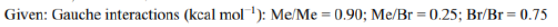Correct option is D
In organic chemistry, the anomeric effect is a stereoelectronic effect that describes the tendency of heteroatomic substituents adjacent to the heteroatom in the ring in, e.g., tetrahydropyran to prefer the axial orientation instead of the less-hindered equatorial orientation that would be expected from steric considerations.
The name comes from the term used to designate the lowest-numbered ring carbon of a pyranose, the anomeric carbon. Isomers that differ only in the configuration at the anomeric carbon are called anomers. The anomers of D-glucopyranose are diastereomers, with the beta anomer having a hydroxyl (−OH) group pointing up equatorially, and the alpha anomer having that (−OH) group pointing down axially.
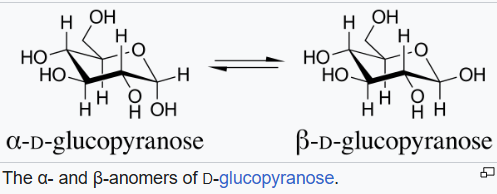
The anomeric effect can also be generalized to any cyclohexyl or linear system with the general formula C−Y−C−X, where Y is a heteroatom with one or more lone pairs, and X is an electronegative atom or group.
Physical explanation
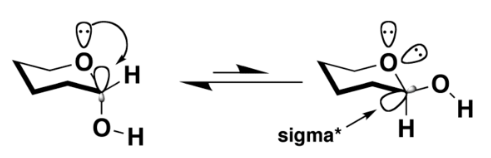
A widely accepted explanation is that there is a stabilizing interaction (hyperconjugation) between the unshared electron pair on the endocyclic heteroatom (within the sugar ring) and the σ* orbital of the axial (exocyclic) C–X bond. This causes the molecule to align the donating lone pair of electrons antiperiplanar (180°) to the exocyclic C-X σ bond, lowering the overall energy of the system and causing more stability.