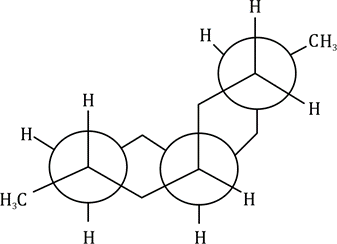
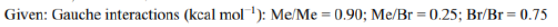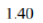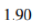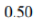Correct option is A
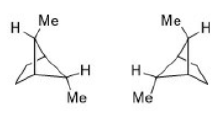
Meso-2,3-dibromobutane has two bromine atoms and two methyl groups attached to adjacent carbon atoms.
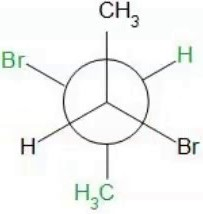
Lowest energy conformer:
For meso-2,3-dibromobutane, this involves anti-positioning the larger groups (e.g., Br/Br and Me/Me)
Next higher energy conformer:
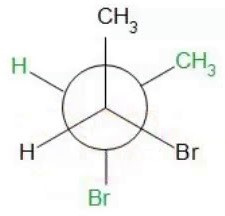
Moving to a higher energy conformer increases the gauche interactions.
Calculations:
Lowest energy conformer: Two Me/Br gauche interactions=0.25+0.25=0.5
Next higher energy conformer:

One Me/Br gauche interaction=0.25

Total energy=0.9+0.25+0.75=1.9
Energy difference=1.9-0.5=1.4 kcal/mol
Information Booster
A Newman projection is a drawing that helps visualize the 3-dimensional structure of a molecule. This projection most commonly sights down a carbon-carbon bond, making it a very useful way to visualize the stereochemistry of alkanes. A Newman projection visualizes the conformation of a chemical bond from front to back, with the front atom represented by the intersection of three lines (a dot) and the back atom as a circle. The front atom is called proximal, while the back atom is called distal.
A staggered projection appears to have the surrounding species equidistant from each other. This kind of conformation tends to experience both anti and gauche interactions. Anti interactions refer to the molecules (usually of the same type) sitting exactly opposite of each other at 180° on the Newman projection. Gauche interactions refer to molecules (also usually of the same type) being 60° from each other on a Newman projection. Anti interactions experience less steric strain than gauche interactions, but both experience less steric strain than the eclipsed conformation.
An eclipsed projection appears to have the surrounding species almost on top of each other. In reality, these species are in line with each other, but are drawn slightly staggered to help format the projection onto paper. These types of conformations are generally higher in energy due to increased bond strain.