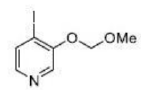
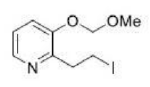
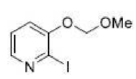
Correct option is B
Organolithiurn reagents react with a wide variety of organic substrates to form carbon-carbon bonds and serve as precursors for the preparation of other organometallic reagents.
Organolithium via Lithium-Hydrogen Exchange (Metalation)
Metal-hydrogen exchange provides a general route to organolithium compounds. The tendency to form the C-Li bond (and thus the reactivity of the C-Li bond) depends on the stability of the R group as a negative ion.
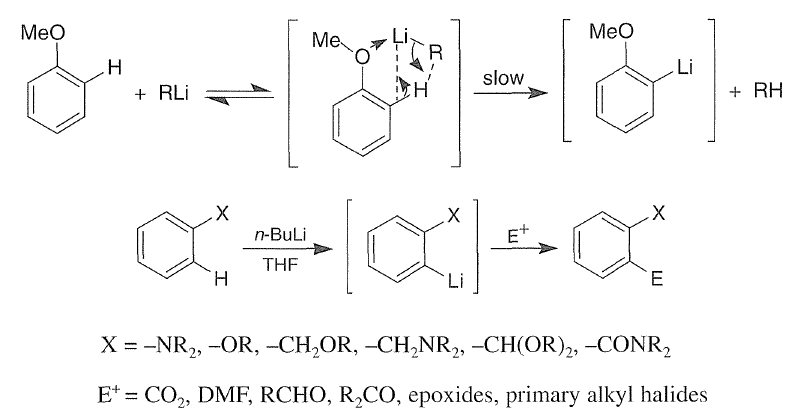
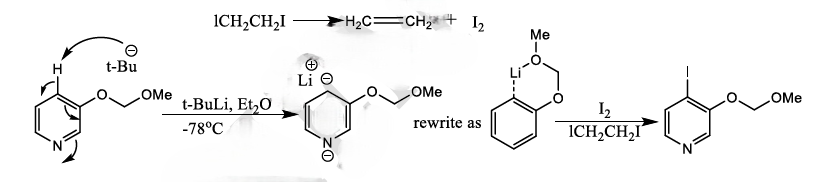
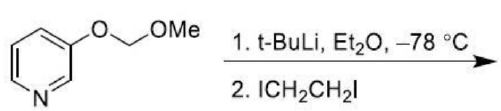
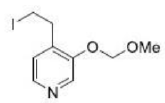
Ortho-Metalation of Substituted Benzenes and Heteroaromatic compounds
Direct metalation of certain aromatic substrates permits regioselective preparation of substituted benzene derivatives and heterocycles. Thus, replacement of a C(sp2)-H by organolithium reagents is facilitated at the ortho-position to a functional group with nonbonding electrons, such as nitrogen or oxygen. Coordination of the lithium reagent with the nitrogen or oxygen holds the organolithium in proximity to the orthohydrogens.