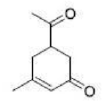
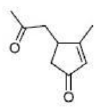
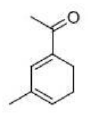
Correct option is B
The aldol reaction is one of the most useful methods for the construction of carbon-carbon bonds. The products of aldol reactions are either β-hydroxy carbonyl compounds or, after dehydration, α, β-unsaturated carbonyl compounds. The aldol reaction is useful not only for making C-C bonds, but also for providing two functional groups, the C=O and a β-OH, which can be further elaborated.
In the strictest sense, the aldol reaction involves the condensation of an enolate derived from an aldehyde or a ketone with another aldehyde or ketone to give a β-hydroxyaldehyde or a β-hydroxyketone, respectively.
Intramolecular Aldol Reaction
The intramolecular aldol condensation is a powerful tool for obtaining five- and six-member rings.
In the following base-catalyzed intramolecular aldol reactions, both enolates are formed reversibly. However, cyclization is faster via attack of the enolate at the less hindered carbonyl group.
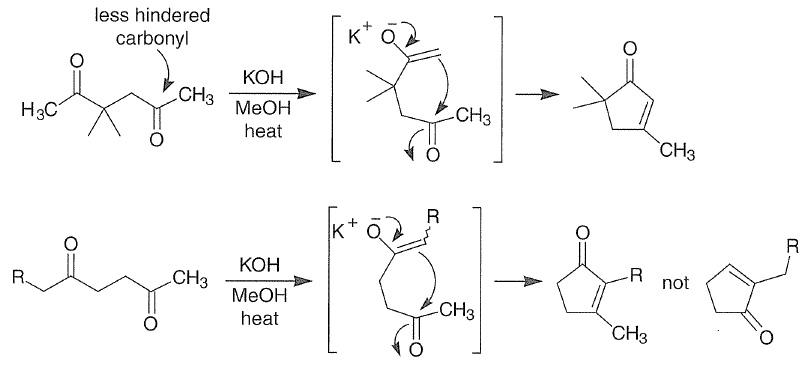
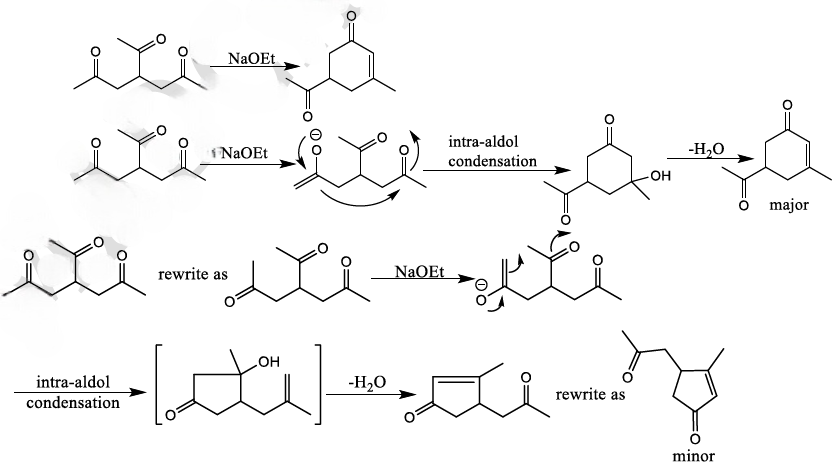
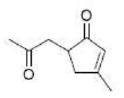
With 1,5-dicarbonyl compounds, two modes of ring closure are often possible. In the example shown below, the more stable (higher-substituted) enone is formed preferentially (thermodynamic control).
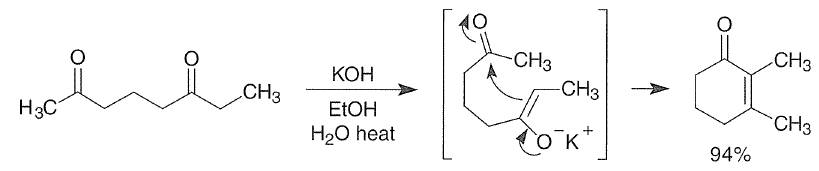
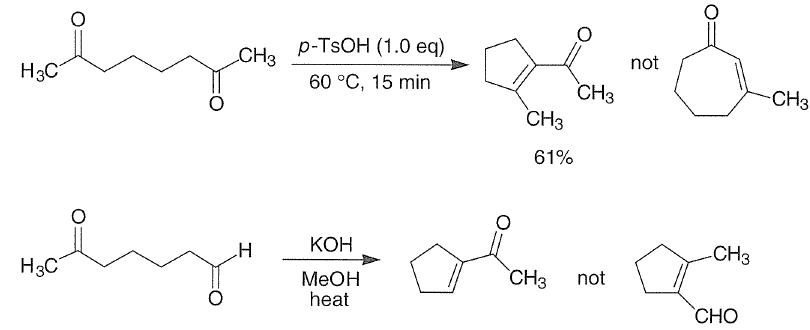
Intramolecular aldol reactions of 1,6-diketones or 1,6-keto-aldehydes afford the corresponding cyclopentenyl carbonyl compounds.