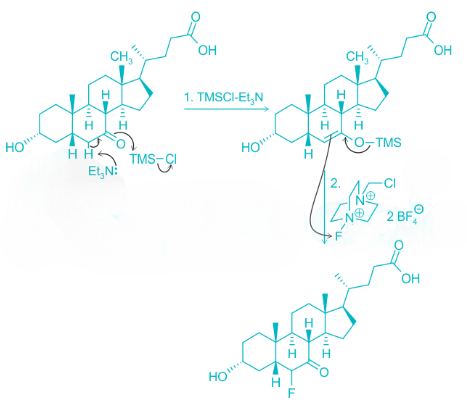
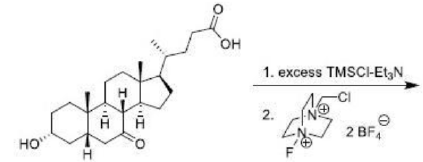
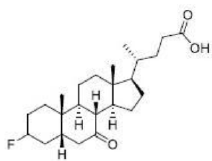
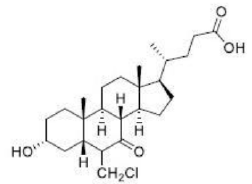
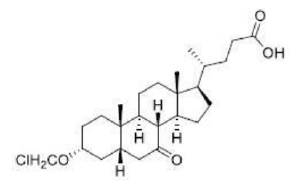
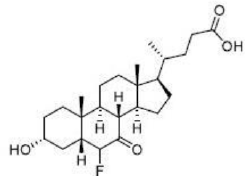
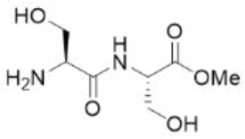
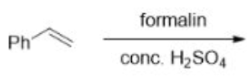Correct option is D
The popularity of silicon protecting groups stems from the fact that they are readily introduced and removed under mild conditions. Moreover, a wide variety of silylating agents are available for tailor-made protection of ROH groups. The chemoselectivity of silylating agents for alcohols and the stability of the resultant silyl ethers toward acid and base hydrolysis, organometallic reagents, and oxidizing and reducing agents increases with increased steric size of the groups attached to silicon. Generally, the sterically least-hindered alcohol is the most readily silylated but is also the most labile to acid or base hydrolysis.
Trimethylsilyl Ethers (RO-TMS)
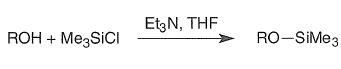
Bases generally employed for the preparation of silyl ethers include R3N, imidazole, DMAP, and DBU (1,8-diazabicyclo[5.4.0]undec-7-ene). Hindered ROH groups are best converted to the corresponding alkoxides with NaH, MeLi, or n-BuLi prior to silylation.
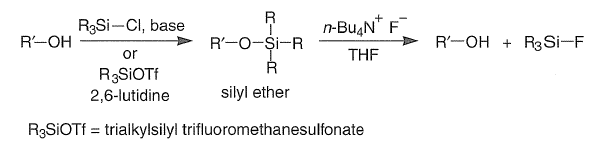
Depending on the structure of silyl ethers, they can be deprotected by H2O, aqueous acids, and fluoride salts. Since silicon has a strong affinity for fluoride ion (bond energy, kcal/mol: Si-F, 143; Si-O), the O-SiR3, bond is especially prone to cleavage by fluoride salts, such as n-Bu4N+F-, which is soluble in organic solvents such as THF and CH2Cl2.