Correct option is A
The reaction of alkenes with ozone constitutes an important method of cleaving carbon-carbon double bonds. Application of low-temperature spectroscopic techniques has provided information about the rather unstable species that are intermediates in the ozonolysis process. These studies, along with isotope labeling results, have provided an understanding of the reaction mechanism. The two key intermediates in ozonolysis are the 1,2,3-trioxolane, or initial ozonide, and the 1,2,4-trioxolane, or ozonide. The first step of the reaction is a cycloaddition to give the 1,2,3-trioxolane. This is followed by a fragmentation and recombination to give the isomeric 1,2,4-trioxolane. The first step is a 1,3-dipolar cycloaddition reaction. Ozone is expected to be a very electrophilic 1,3-dipole because of the accumulation of electronegative oxygen atoms in the ozone molecule. The cycloaddition, fragmentation, and recombination are all predicted to be exothermic on the basis of thermochemical considerations.
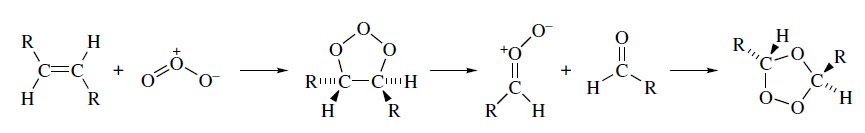
As ozone is electrophilic in nature, it will react with nucleophilic species.
A is most nucleophilic due to the +M effect of the -OMe group.
The +M effect, also known as the positive mesomeric effect, occurs when the substituent is an electron donating group. The group must have one of two things: a lone pair of electrons, or a negative charge. In the +M effect, the pi electrons are transferred from the group towards the conjugate system, increasing the density of the system. Due to the increase in electron density, the conjugate system will develop a more negative charge. As a result, the system under the +M effect will be more reactive towards electrophiles, which can take away the negative charge, than a nucleophile.

C has four Me groups while B has two Me groups. C will be more reactive than B due to the +I effect of four Me groups.
The inductive effect in a molecule is a local change in the electron density due to electron-withdrawing or electron-donating groups elsewhere in the molecule, resulting in a permanent dipole in a bond. It is present in a σ (sigma) bond. Groups having +I effect (Inductive effect) attached to a molecule increases the overall electron density on the molecule and the molecule is able to donate electrons, making it basic.
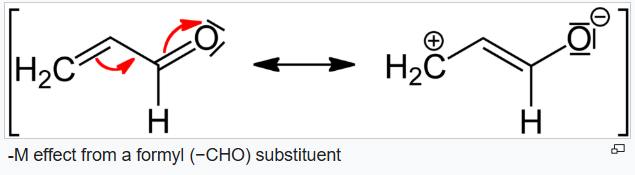
D will be least reactive due to -M effect of the carbonyl group.
The -M effect, also known as the negative mesomeric effect, occurs when the substituent is an electron-withdrawing group. In order for a negative mesomeric (-M) effect to occur the group must have a positive charge or an empty orbital in order to draw the electrons towards it. In the -M effect, the pi electrons move away from the conjugate system and towards the electron drawing group. In the conjugate system, the density of electrons decreases and the overall charge becomes more positive. With the -M effect the groups and compounds become less reactive towards electrophiles, and more reactive toward nucleophiles, which can give up electrons and balance out the positive charge.