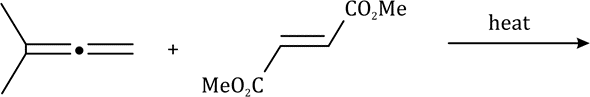Correct option is A
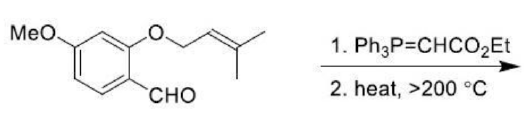
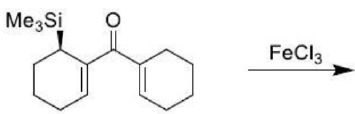
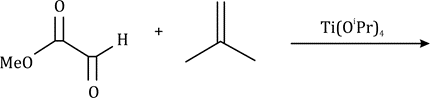
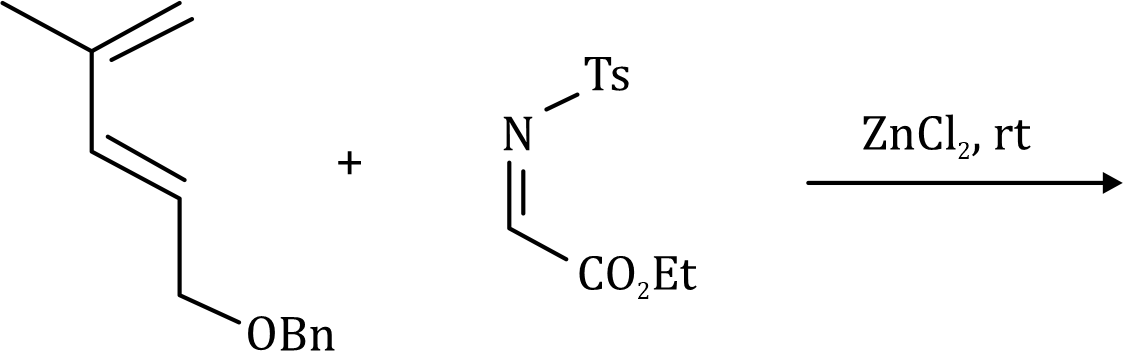
The Claisen rearrangement is a powerful carbon–carbon bond-forming chemical reaction. The heating of an allyl vinyl ether will initiate a [3,3]-sigmatropic rearrangement to give a γ,δ-unsaturated carbonyl.
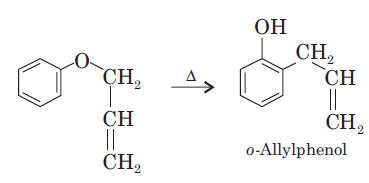
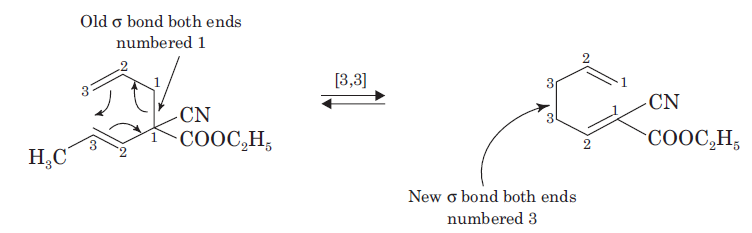
The above Claisen rearrangement is two step reaction. The first step in this reaction is [3, 3] sigmatropic rearrangement.
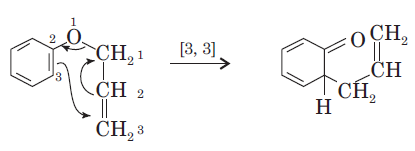
This is one step mechanism without ionic intermediates. In this case numbering start from the heteroatom oxygen having σ (sigma) bond and allylic carbon of the allyl group. The second step in the reaction is a simple ionic proton transfer to regenerate aromaticity.
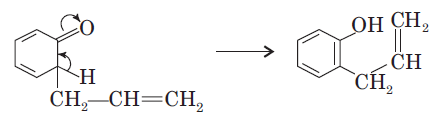
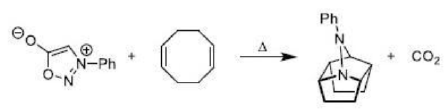
Cope rearrangement
The thermal rearrangement of 1, 5-dienes by [3, 3] sigmatropy is called Cope rearrangement. The reaction proceeds in the thermodynamically favoured direction.