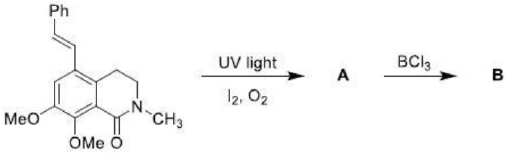
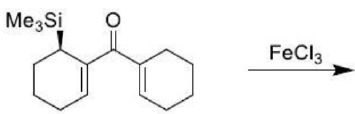
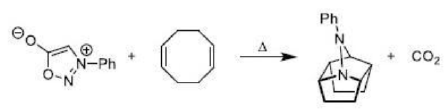
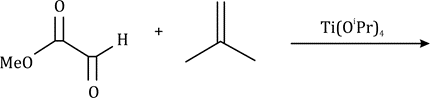
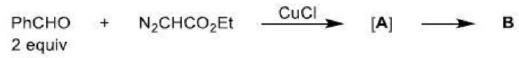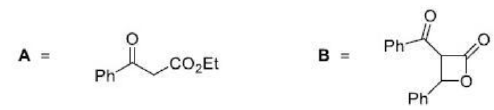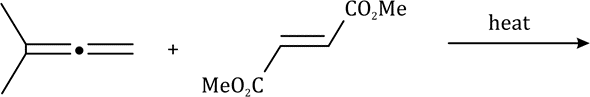Correct option is A
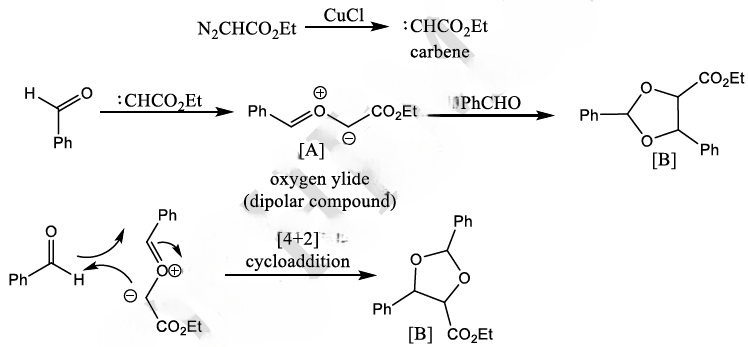
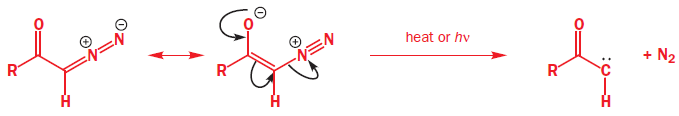
Diazocarbonyl compound are stable, because the electron-withdrawing carbonyl group stabilizes the diazo dipole, and are very useful sources of carbenes carrying a carbonyl substituent.
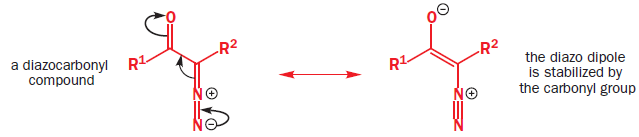
Diazocarbonyl compounds can be decomposed to carbenes by heat or light. The formation of very stable gaseous nitrogen compensates for the formation of the unstable carbene.
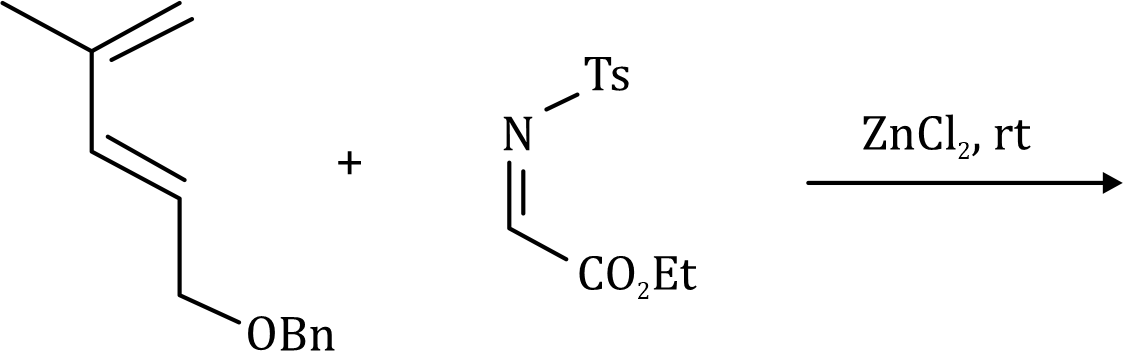
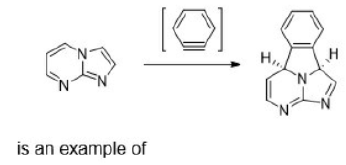
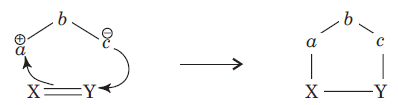
The 1,3-dipolar cycloaddition is a chemical reaction between a 1,3-dipole and a dipolarophile to form a five-membered ring.
The species
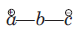
is called the 1, 3-dipolar molecule and X=Y is the dipolarophile.

In 1, 3-dipolar species,
a has six electrons in its outermost orbit. b has its complete octet having at least one lone pair of electrons. c has its complete octet having negative charge.
(ii) a may be carbon, oxygen or nitrogen, b may be nitrogen or oxygen, and c may be carbon, oxygen or nitrogen.
(iii) If b is nitrogen then it has single or double bond. If b is oxygen then it has single bond only.
The dipolarophiles (X=Y) are alkenes, alkynes, imines, nitriles and carbonyl compounds.