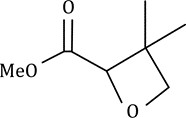
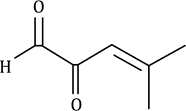
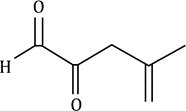
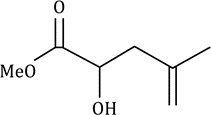
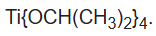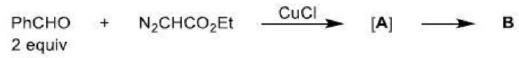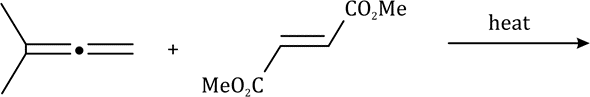Correct option is D
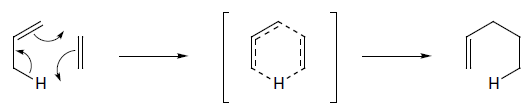
The ene reaction
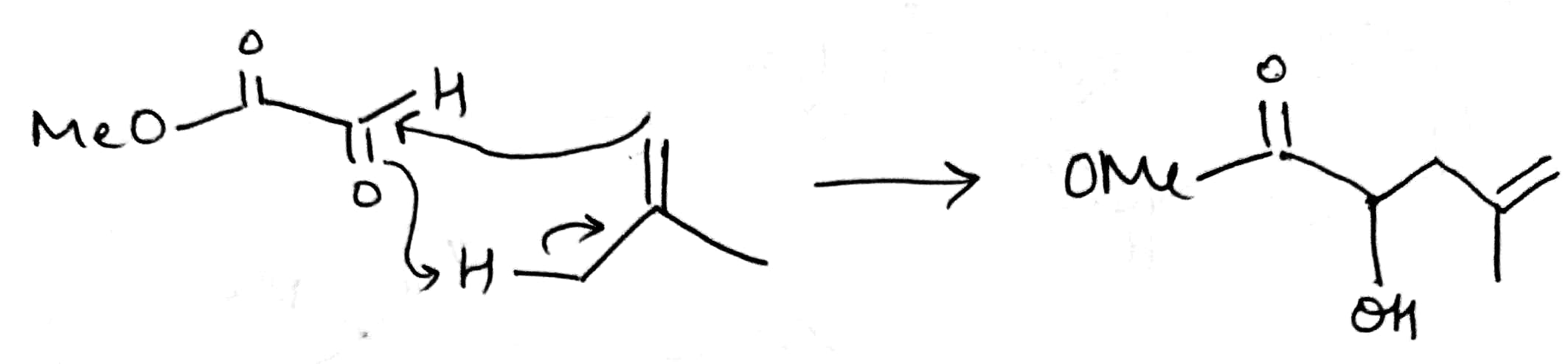
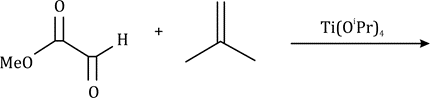
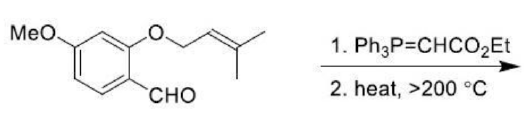
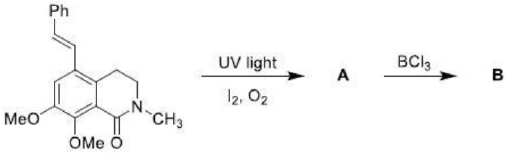
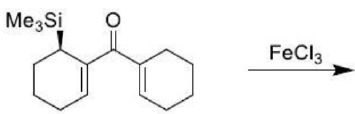
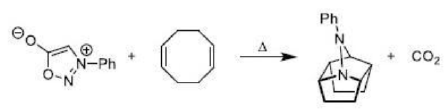
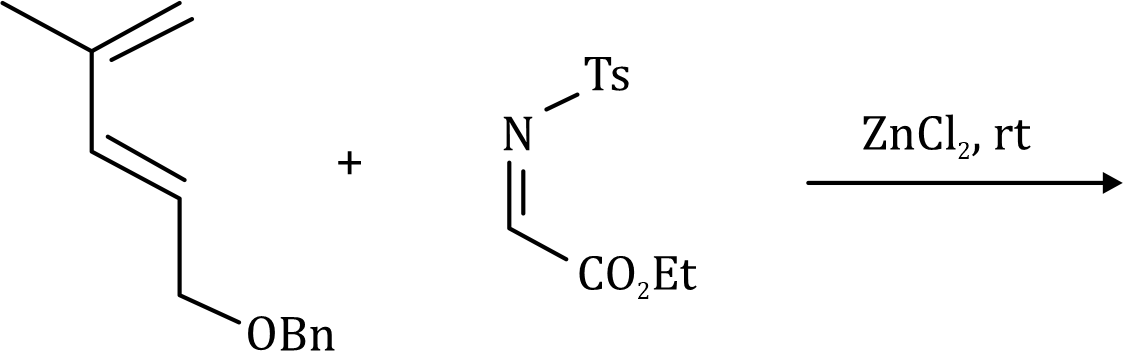
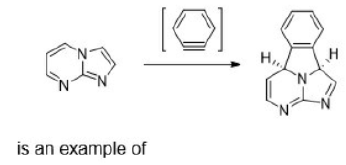
The ene reaction involves the thermal reaction of an alkene bearing an allylic hydrogen atom (the ene component) with a compound containing an activated multiple bond (the enophile). A new sigma-bond is formed between the unsaturated centres with migration of the allylic hydrogen atom to the other terminus of the enophile multiple bond.
Many ene reactions are catalysed by Lewis acids and proceed under relatively mild conditions, often with improved stereoselectivity. It is possible to invoke frontier orbital theory to explain these reactions, with the Lewis acid catalyst exerting its effect by lowering the energy of the LUMO of the enophile.
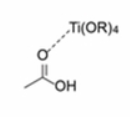
Titanium(IV) compounds, especially titanium alkoxides, are often employed as esterification catalyst due to their inherent Lewis acidity and non-toxic nature.
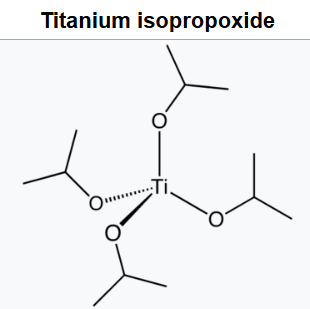
Titanium isopropoxide, also commonly referred to as titanium tetraisopropoxide or TTIP, is a chemical compound with the formula