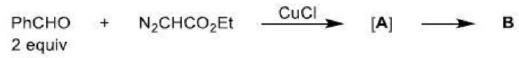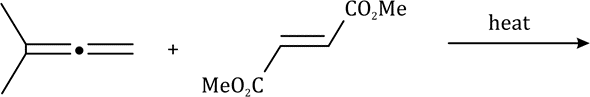Correct option is B
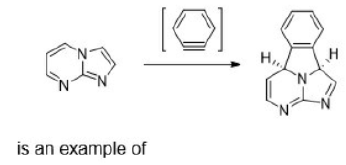
In organic chemistry, a cycloaddition is a chemical reaction in which "two or more unsaturated molecules (or parts of the same molecule) combine with the formation of a cyclic adduct in which there is a net reduction of the bond multiplicity". The resulting reaction is a cyclization reaction. Cycloadditions can be described using two systems of notation. An older but still common notation is based on the size of linear arrangements of atoms in the reactants. It uses parentheses: (i + j + …) where the variables are the numbers of linear atoms in each reactant. The product is a cycle of size (i + j + …). In this system, the standard Diels-Alder reaction is a (4 + 2)-cycloaddition, the 1,3-dipolar cycloaddition is a (3 + 2)-cycloaddition and cyclopropanation of a carbene with an alkene a (2 + 1)-cycloaddition.
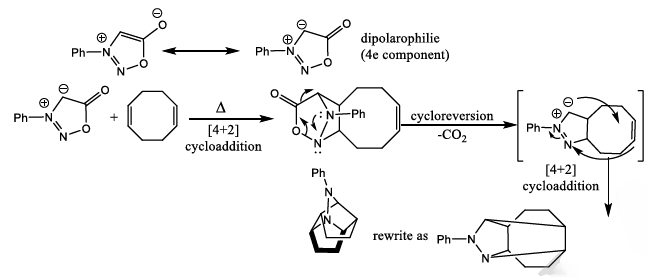
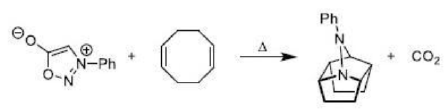
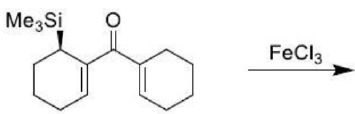
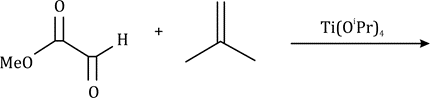
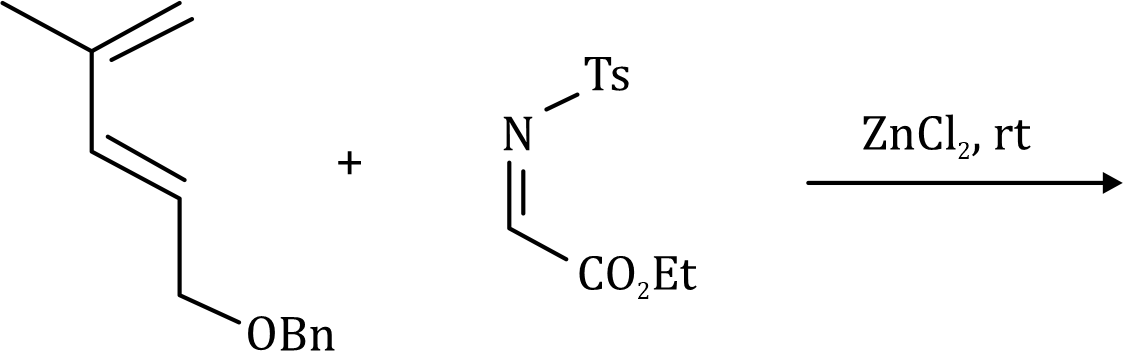
There is a large class of reactions, known as 1, 3-dipolar cycloaddition reactions. The species
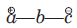
is called the 1, 3-dipolar molecule and X=Y is the dipolarophile.
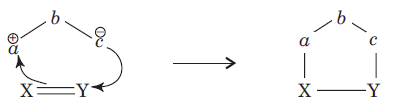
In 1, 3-dipolar species
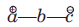
(i) a has six electrons in its outermost orbit. b has its complete octet having at least one lone pair of electrons. c has its complete octet having negative charge.
(ii) a may be carbon, oxygen or nitrogen, b may be nitrogen or oxygen, and c may be carbon, oxygen or nitrogen.
(iii) If b is nitrogen then it has single or double bond. If b is oxygen then it has single bond only.
The dipolarophiles (X=Y) are alkenes, alkynes, imines, nitriles and carbonyl compounds.
Cycloreversion can be described as the reversal of cycloaddition, i.e. conversion of a cyclic compound to its acyclic constituents, from which it is (at least formally) derived. It is a thermal process.